Specialist Fund Concentration in DMD Treatments: Pipeline and Mechanisms
Duchenne muscular dystrophy has attracted an unusual concentration of specialist biotech hedge fund interest. We mapped interventional trials across 10 companies and overlaid beneficial ownership data from 18 specialist funds to show where that consensus sits and why.
RxDataLab tracks 18 biotech specialist hedge funds that invest primarily in life sciences focused companies. Monitoring these funds allows us to track new ideas and shifts in interest from some of the most successful investors in science.
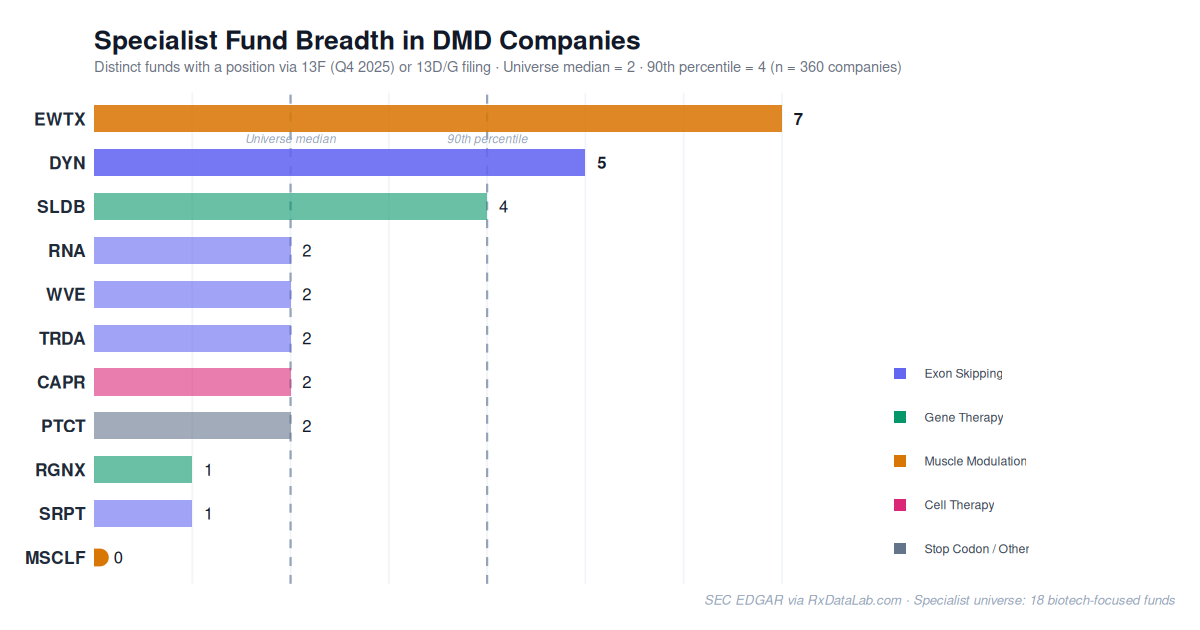
Strategies among these investors varies, but often converges on companies and themes. Currently 360 public companies are held by at least one of these funds — the median is held by 2, and the 90th percentile by 4. Edgewise Therapeutics, a company with a ~$3B market cap, is held by 7. Dyne Therapeutics, also ~$3B market cap, is held by 5. As of April 2026, both are pre-approval and focused on treatments for Duchenne muscular dystrophy (DMD), a rare monogenic disease affecting approximately 1 in 5,000 male births in the US1. Here, we looked closer at this unusual concentration of specialist interest in DMD, including the companies involved and mechanism clusters.
Specialist Fund Consensus Investments in DMD Companies
Count of 18 tracked specialist biotech funds with a position via 13F (Q4 2025) or 13D/G filings (most recent).
DMD Clinical Trial Landscape #
To identify the companies active in DMD and categorize intervention methods, we mapped trials to both the MONDO disease ontology and the NCI Thesaurus (NCIt), identifying 10 companies with active DMD trials across 4 different intervention strategies2. Commercial companies with active trials can be grouped into four clusters based on approach:
- Exon skipping
- Gene therapy
- Muscle modulation
- Cell Therapy
Companies with Active DMD Programs by Mechanism and Phase
Bubble size represents the number of specialist funds with positions (13F or 13D/G) as of Q4 2025.
Exon skipping
Exon skipping is the most populated cluster, with five companies running active trials. The approach uses antisense oligonucleotides (ASO) or antibody oligonucleotide conjugates (AOC) to skip damaged exons to restore the reading frames and allow patients to produce a partially functional dystrophin3. The approach varies in effectiveness based on the patient’s particular mutations and delivery mechanisms, but it is the most de-risked mechanism in the field since Sarepta holds FDA approvals for exon 51, 45, and 53. The competing program targets and stages are summarized in the table below, each targets a different patient subset and uses novel delivery mechanisms.
| Company | 13F (Q4 2025) | 13D/G |
|---|---|---|
| Dyne (DYN) | Baker Brothers, MPM BioImpact, OrbiMed, Perceptive Advisors | Atlas Ventures 13G/A (4.0%) |
| Wave (WVE) | Baker Brothers, RA Capital | RA Capital 13D/A (17.5%) |
| Avidity (RNA) | OrbiMed, RA Capital | RA Capital 13G/A (5.9%) |
| Entrada (TRDA) | Baker Brothers | 5AM Ventures 13D/A (10.8%) |
| Sarepta (SRPT) | MPM BioImpact | — |
Exon 51 covers ~13% of DMD patients, and Sarepta’s eteplirsen has been approved since 2016. Dyne’s DYNE-251 also targets Exon 51 and uses a delivery mechanism intended to improve skeletal and cardiac muscle uptake. Exon 44 has two programs, with Avidity’s (recently acquired by Novartis) AOC 1044 and Entrada’s ENTR-601-44 (peptide conjugates). Entrada also has a Phase 1/2 for exon 45. Wave’s WVE-N531 targets exon 53.
| Exon 44 (~7%) | Exon 45 (~9%) | Exon 51 (~13%) | Exon 53 (~8%) | |
|---|---|---|---|---|
| Sarepta | — | Approved | Approved | Approved |
| Avidity4 | Ph 2 | — | — | — |
| Entrada | Ph 1/2 | Ph 1/2 | — | — |
| Dyne | — | — | Ph 1/2 | — |
| Wave | — | — | — | Ph 1/2 |
The estimated percentage of DMD patients that specific exon skipping is applicable to indicated in the header. Coverage adopted from Bladen et al. 2015 table 2. Trial phase data from RxDataLab.com and ClinicalTrials.gov as of April 2026.
Beyond targeting specific mutations, these players are competing to solve the “delivery problem” inherent in ASO therapies. More effective delivery of ASO’s to skeletal and cardiac muscle tissue is a major hurdle that could result in rapid development for other exons.
Gene therapy
Gene therapy seeks to permanently address the genetic cause of the disease by delivering a microdystrophin construct, typically via AAV. This approach also has an approved product with Sarepta’s Elevidys, though its commercial trajectory has been fraught. Distribution was suspended by the FDA last summer over three patient deaths linked to acute liver failure before resuming. Solid Biosciences (SGT-003, 4 specialist funds) and RegenxBio (RGX-202, 1 fund) both have late-stage programs still running. Whether the access and pricing difficulties Sarepta has faced are product-specific or structural for the gene therapy category is a central open question.
Cell therapy
Capricor Therapeutics’ Deramiocel (CAP-1002) is the lone active Phase 3 program in cell therapy, using allogeneic cardiosphere-derived cells to specifically address the cardiomyopathy that is a primary cause of mortality in DMD. They previously received a complete response letter from the FDA in July 2025, but after new data submissions and negotiations the FDA has agreed to continue reviewing their application. Capricor has a target PDUFA date for Deramiocel of August 22, 2026.
Muscle modulation
Edgewise Therapeutics is taking a different approach with Sevasemten. Sevasemten is a small molecule that stabilizes skeletal muscle fibers to prevent contraction induced breakdown of dystrophin. Unlike exon skipping, it is mutation-agnostic and theoretically applicable to all genotypes. Active Phase 2 programs in both Becker Muscular Dystrophy (GRAND CANYON, n=244) and DMD (LYNX).
Edgewise stands out both due to their unique approach and their significant biotech specialist consensus via ownership stakes. Collectively, four specialist biotech hedge funds own ~36% of the company, with 3 others holding large stakes disclosed via 13F:
- OrbiMed 13D 14.5%
- RA Capital 13G 9.9%
- Baker Brothers Advisors 13G 6.9%
- Perceptive Advisors 13G 4.4%
- Cormorant Asset Management, MPM BioImpact, Deerfield Management all with 13F disclosures.
The other company targeting muscular modulation for DMD is Satellos. Satellos’s approach focuses on muscle regeneration with their small molecule SAT-3247. They aim to restore innate muscle repair by correcting the polarity of satellite cells. Surprisingly, as of April 2026, we do not have records of specialist funds that we track holding Satellos.
What to Watch #
- Edgewise sevasemten Phase 2 readouts. GRAND CANYON (Becker MD, n=244) will be a pivotal test of the mutation-agnostic muscle modulation hypothesis. Following positive 3.5-year data reported in March 2026, a positive signal validates the thesis and the field’s broadest specialist consensus.
- Novartis (formerly Avidity) AOC 1044 Phase 2. Now under the Novartis umbrella as of February 2026, the focus shifts to whether the AOC platform can deliver superior dystrophin restoration. If it does, Novartis will have a powerful RNA-targeting platform to deploy.
- Solid Bio SGT-003 Phase 3 (IMPACT DUCHENNE). Given the challenges with Sarepta’s approved gene therapy, Solid Bio’s trial is a pivotal moment for the gene-therapy approach to DMD. They recently announced positive data from Phase ½ INSPIRE DUCHENNE trial and have FDA alignment on the Phase 3 IMPACT DUCHENNE trial design with plans to discuss accelerated approval pathways with FDA5.
- Dyne DYNE-251 Phase 1/2 data. The 5-fund specialist breadth behind Dyne is a strong vote of confidence in the exon skipping approach and Dyne’s delivery mechanism. In December 2025 they announced positive topline results from their DELIVER trial. Clinical dystrophin production data vs. eteplirsen benchmarks will be the first test of whether that conviction is justified, but the company announced plans to file for accelerated approval in Q2 2026.
RxDataLab maintains a comprehensive biotech industry intelligence database where we track clinical trials, institutional ownership, SEC filings, and more across 500+ biotech companies. See BioHedge for live specialist fund tracking, Strategic Signals for continuous monitoring of biotech SEC filings and the full platform for trial landscape access.
Tracked Specialist Funds #
RxDataLab tracks a subset of biotech specialist funds based on filing patterns. For the purpose of this article, here are the funds:
- 5AM Venture
- ARCH Venture Partners
- Atlas Venture
- Bain Capital Life Sciences
- Baker Brothers Advisors
- BIOS Partners
- BVF Partners
- Cavu Advisors
- Cormorant Asset Management
- Deerfield Management
- EcoR1 Capital
- Fairmount Funds Management
- MPM BioImpact
- OrbiMed Advisors
- Perceptive Advisors
- RA Capital Management
- Redmile Group
- Third Rock Ventures
We are always updating and refining our list of specialists. Reach out if you have one you’d like us to follow.
Muscular Dystrophy Association. https://www.mda.org/disease/duchenne-muscular-dystrophy ↩︎
We filtered for only Industry sponsored active trials. ↩︎
For more information about the biology of exon skipping, see this review from Aslesh et al. https://pmc.ncbi.nlm.nih.gov/articles/PMC5874658 ↩︎
Avidity was acquired by Novartis in a deal that closed in February 2026. ↩︎
See Solid’s recent 8-K filing ↩︎
RxDataLab Research Notes
Primary-source analysis of biotech companies: clinical trials, SEC filings, and hedge fund positioning. Get notified when we publish. No fixed schedule, no filler.