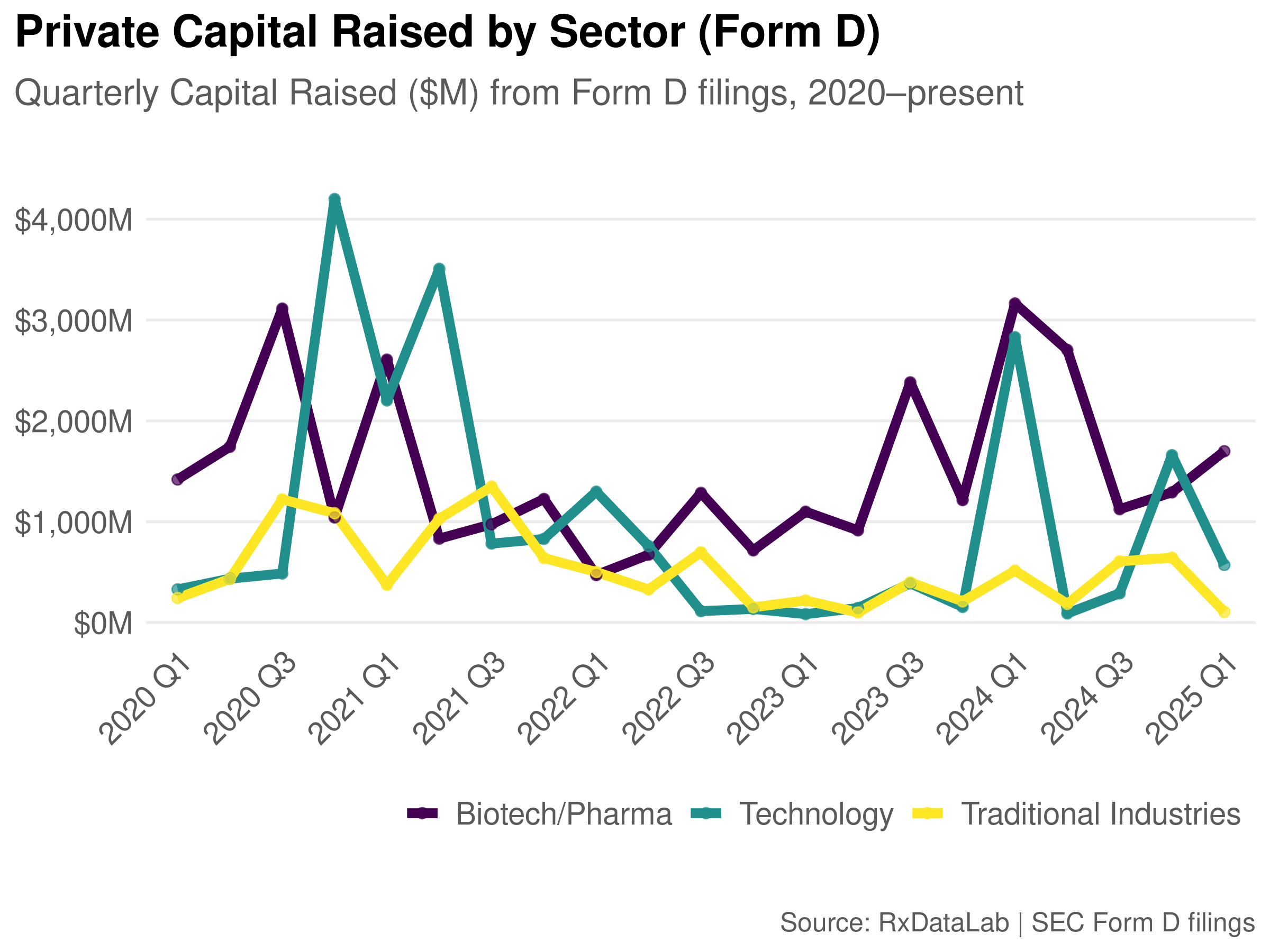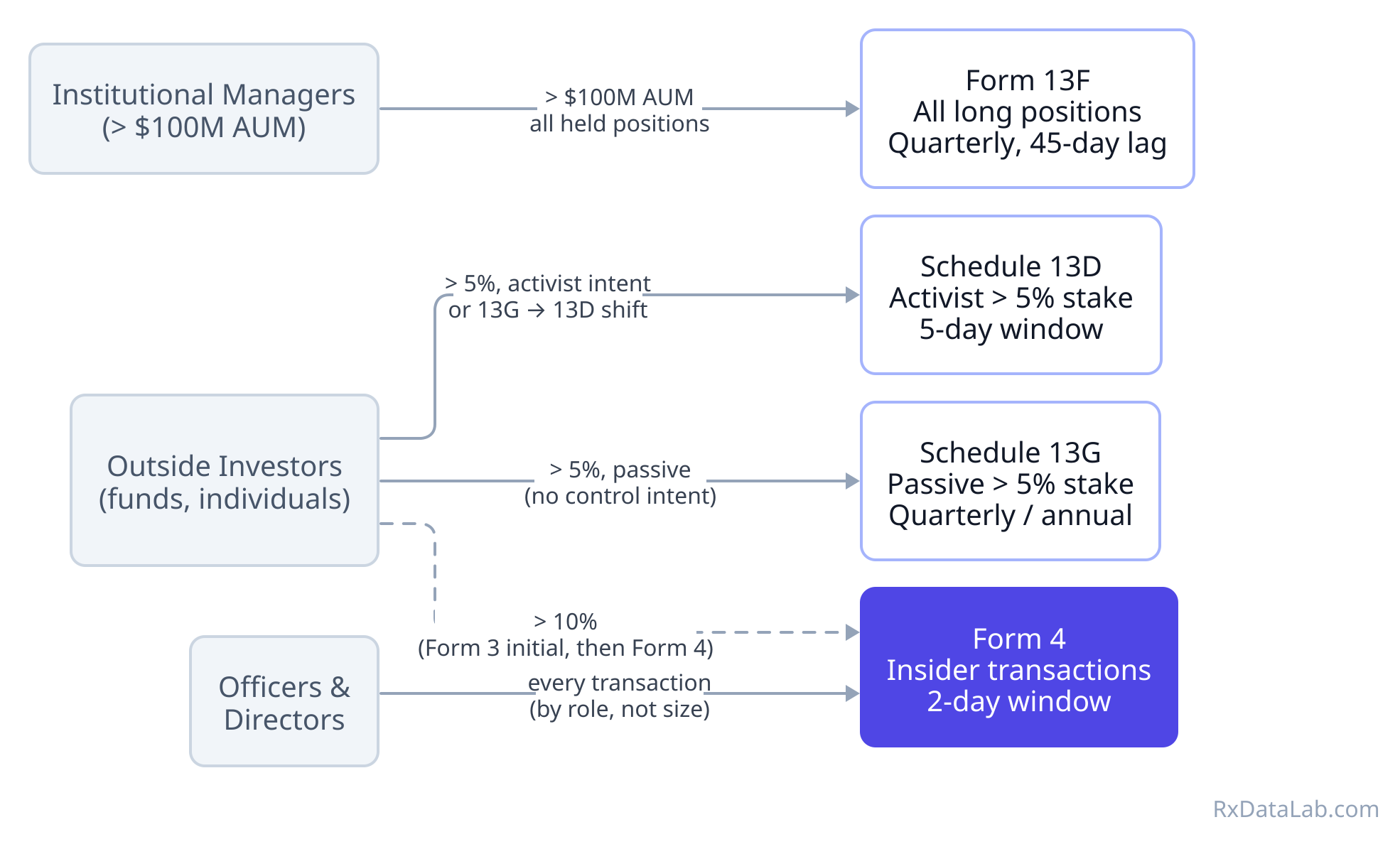
SEC Ownership Filings in Biotech and What We Can Learn From Them
How to read SEC beneficial ownership and insider reporting filings such as Schedule 13D, 13G, Form 13F, and Form 4 and what they reveal when you layer them together for biotech companies.