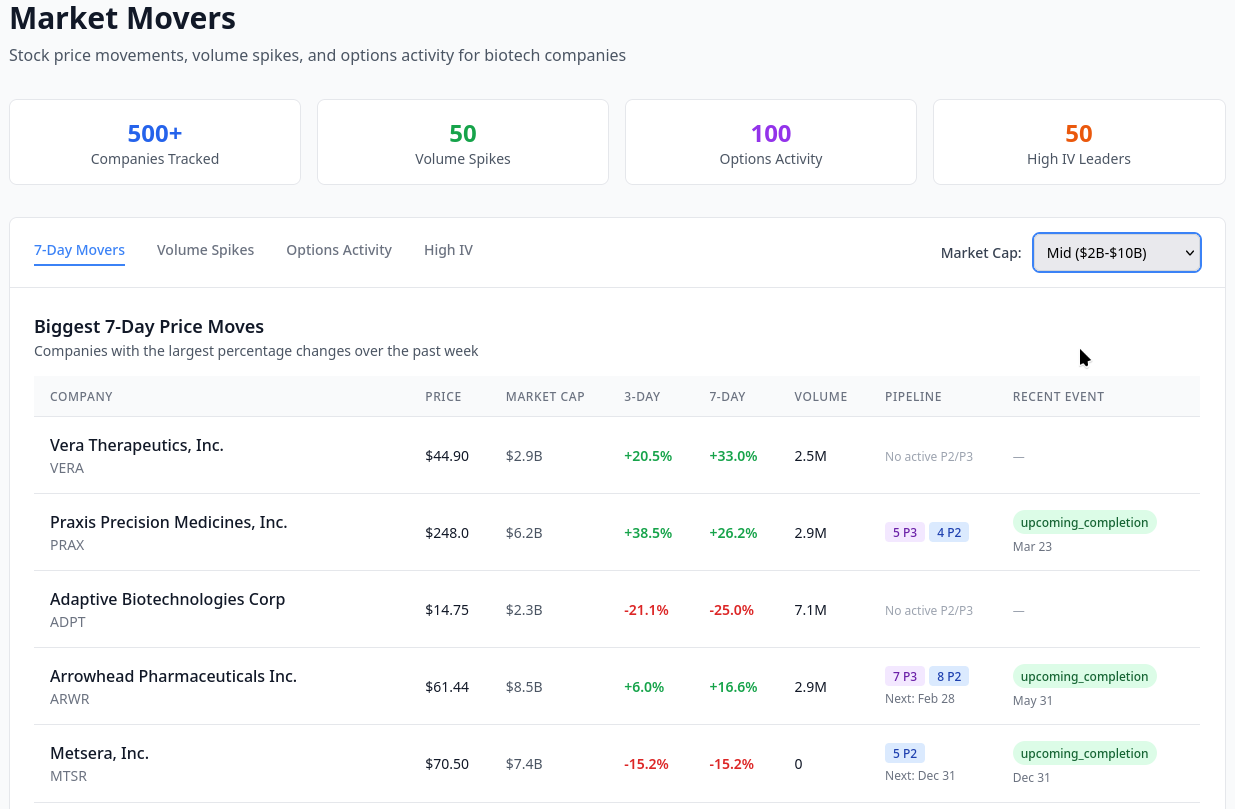
Clinical Trials Dashboard
This dashboard is a work in progress. Is there other information you'd like to see? Are we missing something? Send us an email!
Studies Ending October-November
SK Bioscience Co., Ltd.
A Trial to Assess the Safety, Immunogenicity and Efficacy of a Trivalent Rotavirus P2-VP8 Subunit Vaccine in Prevention of Severe Rotavirus Gastroenteritis in Healthy Infants in Africa and India
Completion: Dec 15, 2025
Phase 3
NCT04010448
Rotavirus Infection of Children
Gala Therapeutics, Inc.
RheOx Registry Study in Europe
Completion: Dec 15, 2025
Phase not applicable
NCT04182841
Chronic Bronchitis
Clinical Trial Selection
| Trial Info | Status and Timeline | Type and Treatment | Condition |
|---|---|---|---|
Pancreatic Cancer Molecular Sub-classification Using Endoscopic Ultrasound Tissue Core Biopsy Samples
Medtronic | Phase not applicable Past estimated: Jun 2, 2024 | No interventions listed | Pancreas Cancer |
Post Market Surveillance of the HeartMate 3 Left Ventricular Assist System in Korea
Abbott Medical Devices | Phase not applicable Completed Jun 2, 2024 | INTERVENTIONAL
| Advanced Heart Failure |
Prizvalve® Transcatheter Valve-in-Valve Implantation Exploratory Study
Shanghai NewMed Medical Co., Ltd. | Recruiting
Phase not applicable Past estimated: Jun 2, 2024 | INTERVENTIONAL
| Failing Bioprosthetic Valve |
Study of Intramuscular Injections of ABBV-950 to Assess Adverse Events and Change in Disease Activity in Adult Participants With Upper Limb Spasticity
AbbVie | Phase 1 Completed Jun 2, 2024 | INTERVENTIONAL
| Upper Limb Spasticity |
Phase I Clinical Trial of CG2001 in Chinese Adult Male Participants With Androgenetic Alopecia
Beijing Dayspring Pharmaceutical Technology Co., Ltd | Phase 1 Completed Jun 2, 2024 | INTERVENTIONAL
| Androgenetic Alopecia (AGA)
Male Pattern of Hair Loss, Androgenic Alopecia |
Study to Assess the Effect of AZD9291 on the Blood Levels of Simvastatin in Patients With EGFRm+ NSCLC
AstraZeneca | Phase 1 Completed Jun 3, 2024 | INTERVENTIONAL
| Non Small Cell Lung Cancer |
A Study to Evaluate 3 Dose Schedules of Daratumumab in Participants With Smoldering Multiple Myeloma
Janssen Research & Development, LLC | Phase 2 Completed Jun 3, 2024 | INTERVENTIONAL
| Multiple Myeloma |
Study of ACY-241 Alone and in Combination With Pomalidomide and Dexamethasone in Multiple Myeloma
Celgene | Phase 1 Completed Jun 3, 2024 | INTERVENTIONAL
| Multiple Myeloma |
Phase III Study of Nazartinib (EGF816) Versus Erlotinib/Gefitinib in First-line Locally Advanced / Metastatic NSCLC With EGFR Activating Mutations
Novartis Pharmaceuticals | Phase 3 Past estimated: Jun 3, 2024 | INTERVENTIONAL
| Carcinoma, Non-small Cell Lung |
Effectiveness, Safety, Adherence, and Health-related Quality of Life in HIV-1 Infected Adults Receiving Bictegravir/ Emtricitabine/Tenofovir Alafenamide
Gilead Sciences | Phase not applicable Completed Jun 3, 2024 | OBSERVATIONAL
| HIV-1-infection |
VT-EBV-N for Treatment of Severe in EBV Positive Extranodal NK/T Cell Lymphoma Patients
ViGenCell Inc. | Recruiting
Phase 2 Past estimated: Jun 3, 2024 | INTERVENTIONAL
| Extranodal NK/T-cell Lymphoma |
Serum Neutrophil Gelatinase-associated Lipocalins (NGAL) and Chronic Kidney Disease
BioPorto Diagnostics | Phase not applicable Completed Jun 3, 2024 | No interventions listed | AKI
Acute Kidney Injury
CKD
Chronic Kidney Diseases
Emergency Department
NGAL
Neutrophil Gelatinase-associated Lipocalin |
CNS Penetration, PK and PD of Preoperative CC-90010 in Progressive/Recurrent Diffuse Astrocytoma, Anaplastic Astrocytoma and Glioblastoma
Celgene | Phase 1 Completed Jun 3, 2024 | INTERVENTIONAL
| Astrocytoma
Glioblastoma |
Safety and Efficacy Study Using Gene Therapy for Critical Limb Ischemia (NL003-CLI-III-1)
Beijing Northland Biotech. Co., Ltd. | Phase 3 Completed Jun 3, 2024 | INTERVENTIONAL
| Arterial Occlusive Disease
Ischemia
Peripheral Vascular Disease
Ulcers |
A Study to Investigate the Efficacy and Safety of Atezolizumab (Tecentriq) in Previously-Treated Patients With Advanced Thymic Carcinoma
Hoffmann-La Roche | Phase 2 Completed Jun 3, 2024 | INTERVENTIONAL
| Carcinoma, Thymic |
In Vitro Study of the Biological and Immunological Activity of Imotopes® Candidates on Blood Cells of Patients With Stabilized NMO
Imcyse SA | Phase not applicable Completed Jun 3, 2024 | INTERVENTIONAL
| Neuromyelitis Optica |
Sym021 in Combination With Either Sym022 or Sym023 or Sym023 and Irinotecan in Patients With Recurrent Advanced Selected Solid Tumor Malignancies
Symphogen A/S | Phase 1 Completed Jun 3, 2024 | INTERVENTIONAL
| Metastatic Cancer
Solid Tumor |
Probiotic Intervention for Gut Function
Probi AB | Phase not applicable Completed Jun 3, 2024 | INTERVENTIONAL
| Gut Function |
REINVENT Registry (Registry of the Nerve Gap Repair From Integra)
Integra LifeSciences Corporation | Phase not applicable Completed Jun 3, 2024 | OBSERVATIONAL
| Peripheral Nerve Injuries |
A Study to Evaluate Safety and Efficacy of AMB-05X Injections in Subjects With TGCT
AmMax Bio, Inc. | Phase 2 Completed Jun 3, 2024 | INTERVENTIONAL
| Pigmented Villonodular Synovitis
TGCT
Tenosynovial Giant Cell Tumor |
SMART Stimulation: Precision Neuromodulation of Cognition in Older Adults
Soterix Medical | Phase not applicable Completed Jun 3, 2024 | INTERVENTIONAL
| Aging |
A Research Study to Understand How Oral Semaglutide Works in People With Type 2 Diabetes in India
Novo Nordisk A/S | Phase not applicable Completed Jun 3, 2024 | OBSERVATIONAL
| Diabetes Mellitus, Type 2 |
AI-09 In Subjects With Glabellar Lines, GL-101
Eirion Therapeutics Inc. | Phase 1 / Phase 2 Completed Jun 3, 2024 | INTERVENTIONAL
| Glabellar Frown Lines |
A Study to Assess NEU-411 in Healthy Participants
Neuron23 Inc. | Phase 1 Completed Jun 3, 2024 | INTERVENTIONAL
| Healthy |
Post Market Observational Retrospective Study of Glycar Bovine Pericardial Patch (CIP-003)
GLYCAR SA (Pty) Ltd | Phase not applicable Completed Jun 3, 2024 | OBSERVATIONAL
| Cardiac Anomaly
Cardiac Defect
Pericardial Defect |
A Study to Investigate the Relative Bioavailability and Food Effect of an Oral Capsid Inhibitor Tablet Formulation Compared With Other Oral Tablet Formulations in Male and Female Healthy Participants
ViiV Healthcare | Phase 1 Completed Jun 3, 2024 | INTERVENTIONAL
| HIV Infections |
Personalized Management of Type 2 Diabetes Using AI-Guided (GenAIS TM) Dietary Supplementation
Triangel Scientific | Phase not applicable Completed Jun 3, 2024 | INTERVENTIONAL
| Diabetes |
A Single Group Study to Evaluate the Effects of a pH Balanced Cleanser on Women's Intimate Health.
Citruslabs | Phase not applicable Completed Jun 3, 2024 | INTERVENTIONAL
| Vaginal Health
Vaginal pH |
A Single Group Study to Evaluate the Effects of a pH Balanced Cleanser on Women's Intimate Health.
Love Wellness | Phase not applicable Completed Jun 3, 2024 | INTERVENTIONAL
| Vaginal Health
Vaginal pH |
Multistrain Probiotics Against Bacterial Vaginosis
Institut AllergoSan Pharma GmbH | Phase 1 Completed Jun 3, 2024 | INTERVENTIONAL
| Bacterial Vaginosis (BV) |
Efficacy and Safety Study of First-line Treatment With Pembrolizumab (MK-3475) Plus Chemotherapy Versus Placebo Plus Chemotherapy in Women With Persistent, Recurrent, or Metastatic Cervical Cancer (MK-3475-826/KEYNOTE-826)
Merck Sharp & Dohme LLC | Phase 3 Completed Jun 4, 2024 | INTERVENTIONAL
| Cervical Cancer |
Study of STRO-002, an Anti-Folate Receptor Alpha (FolRα) Antibody Drug Conjugate in Ovarian & Endometrial Cancers
Sutro Biopharma, Inc. | Phase 1 Completed Jun 4, 2024 | INTERVENTIONAL
| Endometrial Cancer
Endometrioid Adenocarcinoma
Fallopian Tube Cancer
Ovarian Cancer
Ovarian Carcinoma
Ovary Cancer
Primary Peritoneal Carcinoma |
Study of Safety and Efficacy of CFZ533 in Type 1 Diabetes Pediatric and Young Adult Subjects
Novartis Pharmaceuticals | Phase 2 Completed Jun 4, 2024 | INTERVENTIONAL
| Type 1 Diabetes Mellitus |
A Study to Evaluate the Efficacy and Safety of Dapirolizumab Pegol in Study Participants With Moderately to Severely Active Systemic Lupus Erythematosus
UCB Biopharma SRL | Phase 3 Completed Jun 4, 2024 | INTERVENTIONAL
| Systemic Lupus Erythematosus |
Non-comparative Study of IFX-1 Alone or IFX-1+Pembrolizumab in Patients With Locally Advanced or Metastatic cSCC.
InflaRx GmbH | Phase 2 Completed Jun 4, 2024 | INTERVENTIONAL
| SCC - Squamous Cell Carcinoma of Skin |
Non-comparative Study of IFX-1 Alone or IFX-1+Pembrolizumab in Patients With Locally Advanced or Metastatic cSCC.
Merck Sharp & Dohme LLC | Phase 2 Completed Jun 4, 2024 | INTERVENTIONAL
| SCC - Squamous Cell Carcinoma of Skin |
Safety and Efficacy of Allogeneic NK Cell Infusions in Patients With Relapsed/Refractory AML and High Risk MDS
Coeptis Therapeutics | Phase 1 Completed Jun 4, 2024 | INTERVENTIONAL
| AML, Adult Recurrent
MDS |
Study to Evaluate the Safety and Efficacy of CSB-001 Ophthalmic Solution 0.1% in Neurotrophic Keratitis Subjects
Claris Biotherapeutics, Inc. | Phase 1 / Phase 2 Completed Jun 4, 2024 | INTERVENTIONAL
| Neurotrophic Keratitis |
Randomized Double-Blind Phase 2 Efficacy and Safety of Autologous HB-MSCs vs Placebo for Treatment of Multiple Sclerosis
Hope Biosciences Research Foundation | Phase 2 Completed Jun 4, 2024 | INTERVENTIONAL
| Multiple Sclerosis |
A Randomized Controlled Trial of InterVapor® in France - The TARGET Trial
Uptake Medical Technology, Inc. | Phase not applicable Completed Jun 4, 2024 | INTERVENTIONAL
| Emphysema |
A Clinical Trial to Investigate the Safety and Tolerability, Efficacy, Pharmacokinetics, Pharmacodynamics and Immunogenicity of 2 Dose Regimens of ARGX-117 in Adults with Multifocal Motor Neuropathy
argenx | Phase 2 Completed Jun 4, 2024 | INTERVENTIONAL
| Multifocal Motor Neuropathy |
Placebo-controlled, Proof-of-concept Study to Evaluate the Safety and Efficacy of Lanifibranor Alone and in Combination With SGLT2 Inhibitor EmpaGliflozin in patiEnts With NASH and Type 2 Diabetes Mellitus
Inventiva Pharma | Phase 2 Completed Jun 4, 2024 | INTERVENTIONAL
| Diabetes Mellitus, Type 2
NASH - Nonalcoholic Steatohepatitis |
A Phase 2a Study to Investigate REM0046127 in Mild to Moderate Alzheimer's Disease
reMYND | Phase 2 Completed Jun 4, 2024 | INTERVENTIONAL
| Alzheimer Disease |
A Phase 3 Study of Recombinant Anti-IL-17A Humanized Monoclonal Antibody in Chinese Participants With Moderate-to-Severe Plaque Psoriasis
Sunshine Guojian Pharmaceutical (Shanghai) Co., Ltd. | Phase 3 Completed Jun 4, 2024 | INTERVENTIONAL
| Psoriasis |
A Study of Modakafusp Alfa in Adult Participants With Multiple Myeloma
Takeda | Phase 1 Completed Jun 4, 2024 | INTERVENTIONAL
| Multiple Myeloma |
Thumb Hemi-Arthroplasty With Natural Kinematics (THANKS) Trial
Loci Orthopaedics | Phase not applicable Completed Jun 4, 2024 | INTERVENTIONAL
| Osteoarthritis Thumb Base Joint |
Harnessing Allo-immunity to Enhance Immune Checkpoint Inhibitor Responses in Advanced NSCLC
Oncovir, Inc. | Phase 1 / Phase 2 Completed Jun 4, 2024 | INTERVENTIONAL
| Stage IV NSCLC |
A Study Assessing the Mass Balance, Pharmacokinetics, and Metabolite Profiles of a Single Oral Dose of [14C]INCB099280 in Healthy Male Participants
Incyte Corporation | Phase 1 Completed Jun 4, 2024 | INTERVENTIONAL
| Healthy Participants |
A Study to Assess the Bioequivalence Between Brivaracetam Tablet and Dry Syrup in Healthy Japanese Male Study Participants
UCB Biopharma SRL | Phase 1 Completed Jun 4, 2024 | INTERVENTIONAL
| Healthy Study Participants |
A Clinical Study to Evaluate the Tolerance, Pharmacokinetics and Efficacy of TQ-A3334 Tablets
Chia Tai Tianqing Pharmaceutical Group Co., Ltd. | Phase 1 / Phase 2 Completed Jun 4, 2024 | INTERVENTIONAL
| Non Small Cell Lung Cancer |
Clinical Trial of Wrist Ambulatory Blood Pressure Monitor in Conformance With the EN ISO 81060-2:2018+AMD1:2020
Huawei Device Co., Ltd | Phase not applicable Completed Jun 4, 2024 | INTERVENTIONAL
| Blood Pressure |
A Study of Enlicitide Decanoate (MK-0616), Warfarin, and Lisinopril in Healthy Adult Participants (MK-0616-026)
Merck Sharp & Dohme LLC | Phase 1 Completed Jun 4, 2024 | INTERVENTIONAL
| Healthy |
A Phase 2 Study to Evaluate Safety of Long-term AL001 Dosing in Frontotemporal Dementia (FTD) Patients (INFRONT-2)
Alector Inc. | Phase 2 Completed Jun 5, 2024 | INTERVENTIONAL
| Frontotemporal Dementia |
A Phase 2 Study to Evaluate Safety of Long-term AL001 Dosing in Frontotemporal Dementia (FTD) Patients (INFRONT-2)
GlaxoSmithKline | Phase 2 Completed Jun 5, 2024 | INTERVENTIONAL
| Frontotemporal Dementia |
SEMA4D Blockade Safety and Brain Metabolic Activity in Alzheimer's Disease (AD)
Vaccinex Inc. | Phase 1 / Phase 2 Completed Jun 5, 2024 | INTERVENTIONAL
| Alzheimer Disease |
A Study of Mirikizumab (LY3074828) in Participants With Ulcerative Colitis
Eli Lilly and Company | Phase 3 Past estimated: Jun 5, 2024 | INTERVENTIONAL
| Ulcerative Colitis |
Sweet Tooth: Nature or Nurture? Role of Long-term Dietary Sweetness Exposure on Sweetness Preferences
Arla Foods | Phase not applicable Completed Jun 5, 2024 | INTERVENTIONAL
| Food Preferences |
Sweet Tooth: Nature or Nurture? Role of Long-term Dietary Sweetness Exposure on Sweetness Preferences
Cargill | Phase not applicable Completed Jun 5, 2024 | INTERVENTIONAL
| Food Preferences |
Sweet Tooth: Nature or Nurture? Role of Long-term Dietary Sweetness Exposure on Sweetness Preferences
Unilever R&D | Phase not applicable Completed Jun 5, 2024 | INTERVENTIONAL
| Food Preferences |
Study of Irinotecan Liposome Injection in Patients With Advanced Breast Cance
CSPC Ouyi Pharmaceutical Co., Ltd. | Phase 1 Completed Jun 5, 2024 | INTERVENTIONAL
| Advanced Breast Cancer |
A Study to Determine the Safety, Pharmacokinetics, and Pharmacodynamics of DNL343 in Participants With Amyotrophic Lateral Sclerosis
Denali Therapeutics Inc. | Phase 1 Completed Jun 5, 2024 | INTERVENTIONAL
| Amyotrophic Lateral Sclerosis |
Study of the Effectiveness and Safety of SHR-1314 for Psoriatic Arthritis
Suzhou Suncadia Biopharmaceuticals Co., Ltd. | Phase 2 Completed Jun 5, 2024 | INTERVENTIONAL
| Psoriatic Arthritis |
Survey Among Healthcare Professionals Treating Patients With Metastatic Breast Cancer in Selected European Countries to Evaluate Their Knowledge on Management of Hyperglycemia When Using Alpelisib
Novartis Pharmaceuticals | Phase not applicable Completed Jun 5, 2024 | OBSERVATIONAL
| Breast Cancer |
Assess Safety and Tolerability of ART-123 + FOLFOX + Bevacizumab in Metastatic Colorectal Cancer Patients
Veloxis Pharmaceuticals | Phase 1 Completed Jun 5, 2024 | INTERVENTIONAL
| Chemotherapy-induced Peripheral Neuropathy |
A Trial to Assess Steviol Glycosides on Acute Appetite Hormone Release
Cargill | Phase not applicable Completed Jun 5, 2024 | INTERVENTIONAL
| Overweight and Obesity
Type 2 Diabetes Mellitus |
A Study of Nanrilkefusp Alfa (SOT101) in Combination With Cetuximab to Evaluate the Efficacy and Safety in Patients With Colorectal Cancer
SOTIO Biotech AG | Phase 2 Completed Jun 5, 2024 | INTERVENTIONAL
| Colorectal Cancer |
Noninvasive Monitoring of Uterine Activity and Fetal Heart Rate
OB-Tools Ltd. | Phase not applicable Completed Jun 5, 2024 | OBSERVATIONAL
| Fetal Heart Rate or Rhythm Abnormality Affecting Newborn During Labor
Obstetric Complication
Uterine Contractions |
Study of Two Digital Therapeutics for the Prevention of Episodic Migraine Receiving CGRP Therapy (ReMMiD-C)
Click Therapeutics, Inc. | Phase 3 Completed Jun 5, 2024 | INTERVENTIONAL
| Episodic Migraine
Headache
Headache, Migraine
Migraine |
A Study in Healthy People to Test Whether Iclepertin Has an Effect on Cardiac Safety
Boehringer Ingelheim | Phase 1 Completed Jun 5, 2024 | INTERVENTIONAL
| Healthy |
A Retrospective Observational Study of Ustekinumab Among Bio-naive Participants With Crohn's Disease in China
Xian-Janssen Pharmaceutical Ltd. | Phase not applicable Completed Jun 5, 2024 | OBSERVATIONAL
| Crohn Disease |
A Study to Evaluate Efficacy of EB-PA as Protein Amplifier for Skeletal Muscle Strength & Growth
Vedic Lifesciences Pvt. Ltd. | Phase not applicable Past estimated: Jun 5, 2024 | INTERVENTIONAL
| Skeletal Muscle Strength & Growth |
A Study of GZR18 Injection in Obese/Overweight Patients
Gan and Lee Pharmaceuticals, USA | Phase 2 Completed Jun 5, 2024 | INTERVENTIONAL
| Obesity
Overweight |
The COAT Trial: Cequa's Onset of Action Trial
Research Insight LLC | Phase 4 Completed Jun 5, 2024 | INTERVENTIONAL
| Dry Eye |
Efficacy and Safety Study of Aprepitant Injection for Prevention of Post-operative Nausea and Vomiting
Qilu Pharmaceutical (Hainan) Co., Ltd. | Phase 3 Completed Jun 5, 2024 | INTERVENTIONAL
| Postoperative Nausea and Vomiting |
Efficacy Study of Nivolumab Plus Ipilimumab or Nivolumab Plus Chemotherapy Against Chemotherapy in Stomach Cancer or Stomach/Esophagus Junction Cancer
Bristol-Myers Squibb | Phase 3 Completed Jun 6, 2024 | INTERVENTIONAL
| Esophageal Adenocarcinoma
Gastric Cancer
Gastroesophageal Junction Cancer |
Efficacy Study of Nivolumab Plus Ipilimumab or Nivolumab Plus Chemotherapy Against Chemotherapy in Stomach Cancer or Stomach/Esophagus Junction Cancer
Ono Pharmaceutical Co. Ltd | Phase 3 Completed Jun 6, 2024 | INTERVENTIONAL
| Esophageal Adenocarcinoma
Gastric Cancer
Gastroesophageal Junction Cancer |
Post-Market Clinical Follow-up Study with ASPIREX®S to Assess the Safety and Effectiveness in the Treatment of DVT
Straub Medical AG | Phase not applicable Completed Jun 6, 2024 | No interventions listed | Bypass Complication
Deep Vein Thrombosis
Dialysis Shunt
Stent Occlusion |
PROSEEK: A Phase 2 Study In Early Parkinson's Disease Patients Evaluating The Safety And Efficacy Of Abl Tyrosine Kinase Inhibition Using K0706
Sun Pharma Advanced Research Company Limited | Phase 2 Completed Jun 6, 2024 | INTERVENTIONAL
| Early Parkinson Disease |
Study to Learn More About the Safety and Effectiveness of Rivaroxaban (Xarelto) When Given Together With Acetylsalicylic Acid to Indian People With Narrowing of the Arteries of the Heart (CAD) and/or With Reduced Blood Flow in the Arteries of the Legs and Arms With Symptoms (Symptomatic PAD)
Bayer | Phase not applicable Completed Jun 6, 2024 | OBSERVATIONAL
| Coronary Artery Disease (CAD)
Prevention of Atherothrombotic Events
Symptomatic Peripheral Artery Disease (Symptomatic PAD) |
A Study to Test the Safety, Pharmacokinetics, and Efficacy of UCB9741 in Healthy Study Participants and in Study Participants With Atopic Dermatitis
UCB Biopharma SRL | Phase 1 / Phase 2 Completed Jun 6, 2024 | INTERVENTIONAL
| Atopic Dermatitis |
Efficacy and Safety of Lomitapide in Paediatric Patients With Homozygous Familial Hypercholesterolaemia (HoFH)
Amryt Pharma | Phase 3 Completed Jun 6, 2024 | INTERVENTIONAL
| Homozygous Familial Hypercholesterolaemia (HoFH) |
SER150 vs Placebo in Diabetic Kidney Disease
Serodus AS | Phase 2 / Phase 3 Completed Jun 6, 2024 | INTERVENTIONAL
| Diabetic Kidney Disease |
A Study to Assess the Safety and Efficacy of Inclacumab in Participants With Sickle Cell Disease Experiencing Vaso-occlusive Crises
Pfizer | Phase 3 Completed Jun 6, 2024 | INTERVENTIONAL
| Sickle Cell Disease
Vaso-occlusive Crisis
Vaso-occlusive Pain Episode in Sickle Cell Disease |
A Clinical Trial Evaluating the Efficacy and Safety of HSK21542 in Patients with Chronic Kidney Disease-Associated Pruritus
Haisco Pharmaceutical Group Co., Ltd. | Phase 3 Completed Jun 6, 2024 | INTERVENTIONAL
| Hemodialysis Patients with Moderate-to-Severe Pruritus
Pruritus |
Extended Treatment and Follow-up of Subjects Treated With Belumosudil in Study KD025-208 or Study KD025-213
Kadmon, a Sanofi Company | Phase 2 Completed Jun 6, 2024 | INTERVENTIONAL
| Chronic Graft-versus-host-disease |
A Study to Test How Well Different Doses of BI 1815368 Are Tolerated by Healthy Men
Boehringer Ingelheim | Phase 1 Completed Jun 6, 2024 | INTERVENTIONAL
| Healthy |
Evaluation of Home-based Sensor System to Detect Health Decompensation in Elderly Patients With History of CHF or COPD
Sensorum Health Inc. | Recruiting
Phase not applicable Past estimated: Jun 6, 2024 | OBSERVATIONAL
| CHF
COPD
COPD Exacerbation
Chronic Obstructive Pulmonary Disease
Congestive Heart Failure |
Brazilian Reality in the Diagnosis and Treatment of Diffuse Large B Cell Lymphoma - BRA-DLBCL
AstraZeneca | Phase not applicable Completed Jun 6, 2024 | No interventions listed | Diffuse Large B Cell Lymphoma |
Pilot Clinical Investigation to Evaluate a Prototype of Sensor Measuring Ventilator-derived Parameters
Archeon | Phase not applicable Completed Jun 6, 2024 | INTERVENTIONAL
| Mechanically Ventilated Patients |
A Clinical Study to Evaluate the Safety of CS-1103 in Healthy Participants
Clear Scientific, Inc. | Phase 1 Completed Jun 6, 2024 | INTERVENTIONAL
| Methamphetamine Abuse
Methamphetamine Intoxication
Substance Use Disorders |
Xpert Hepatitis-C Virus (HCV) Test on the GeneXpert Xpress System
Cepheid | Phase not applicable Completed Jun 6, 2024 | OBSERVATIONAL
| HCV |
Allogeneic Hematopoietic Stem Cell Transplantation in Patients With Myelodysplastic Syndrome Low Risk
Neovii Biotech | Phase not applicable Completed Jun 7, 2024 | INTERVENTIONAL
| MDS |
Allogeneic Hematopoietic Stem Cell Transplantation in Patients With Myelodysplastic Syndrome Low Risk
Novartis | Phase not applicable Completed Jun 7, 2024 | INTERVENTIONAL
| MDS |
A Study of Pre-Operative Treatment With Cryoablation and Immune Therapy in Early Stage Breast Cancer
Bristol-Myers Squibb | Phase not applicable Completed Jun 7, 2024 | INTERVENTIONAL
| Breast Cancer |
A Study of INCB050465 in Relapsed or Refractory Follicular Lymphoma
Incyte Corporation | Phase 2 Completed Jun 7, 2024 | INTERVENTIONAL
| Lymphoma |
Study of the Efficacy and Safety of HS-10234 in Patients With Chronic Hepatitis B Virus Infection
Jiangsu Hansoh Pharmaceutical Co., Ltd. | Phase 3 Completed Jun 7, 2024 | INTERVENTIONAL
| Chronic HBV Infection |
Improving Neurological Health in Aging Via Neuroplasticity-based Computerized Exercise
Posit Science Corporation | Phase not applicable Completed Jun 7, 2024 | INTERVENTIONAL
| Healthy Aging |
The MOOD Study - External Combined Occipital and Trigeminal Nerve Stimulation (eCOT-NS) for the Treatment of Major Depressive Disorder (MDD)
Neurolief Ltd. | Phase not applicable Completed Jun 7, 2024 | INTERVENTIONAL
| MDD |
Effect of Tumor Treating Fields (TTFields, 150 KHz) Concomitant with Chemotherapy As First Line Treatment of Unresectable Gastroesophageal Junction or Gastric Adenocarcinoma
NovoCure Ltd. | Phase not applicable Completed Jun 7, 2024 | INTERVENTIONAL
| Gastric Cancer
GastroEsophageal Cancer |
Effect of Tumor Treating Fields (TTFields, 150 KHz) Concomitant with Chemotherapy As First Line Treatment of Unresectable Gastroesophageal Junction or Gastric Adenocarcinoma
Zai Lab (Shanghai) Co., Ltd. | Phase not applicable Completed Jun 7, 2024 | INTERVENTIONAL
| Gastric Cancer
GastroEsophageal Cancer |
StrataGraft Overlay of Meshed Autograft in Full-thickness Thermal Burns
Stratatech, a Mallinckrodt Company | Phase 1 / Phase 2 Completed Jun 7, 2024 | INTERVENTIONAL
| Full Thickness Thermal Burn |
Amyloid Prediction in Early Stage Alzheimer's Disease From Acoustic and Linguistic Patterns of Speech - FUTURE Extension
Novoic Limited | Phase not applicable Completed Jun 7, 2024 | No interventions listed | Alzheimer Disease
Alzheimer's Disease (Incl Subtypes)
Mild Cognitive Impairment
Preclinical Alzheimer's Disease
Prodromal Alzheimer's Disease |
A Study to Evaluate ELU001 in Patients With Solid Tumors That Overexpress Folate Receptor Alpha (FRα)
Elucida Oncology | Phase 1 / Phase 2 Completed Jun 7, 2024 | INTERVENTIONAL
| Endometrial Adenocarcinoma
Endometrial Cancer
Endometrial Cancer Recurrent
Endometrial Carcinosarcoma
Endometrial Clear Cell Adenocarcinoma
Endometrial Diseases
Endometrial Neoplasms
Endometrioid Adenocarcinoma
Endometrioid Tumor
Fallopian Tube Cancer
Ovarian Adenocarcinoma
Ovarian Cancer
Ovarian Cancer Recurrent
Ovarian Cancer Stage
Ovarian Carcinoma
Ovarian Diseases
Ovarian Epithelial Cancer
Ovarian Neoplasm Epithelial
Ovarian Neoplasms
Ovarian Serous Adenocarcinoma
Ovary Cancer
Ovary Disease
Ovary Metastasis
Ovary Neoplasm
Peritoneal Cancer |
Evaluation of Psilocybin (TRP-8802) in the Treatment of Binge Eating Disorder
TRYP Therapeutics | Phase 2 Completed Jun 7, 2024 | INTERVENTIONAL
| Binge Eating Disorder |
Physician Acceptance of the NeuTrace System for Cardiac Electroanatomic Mapping
NeuTrace, Inc. | Recruiting
Phase not applicable Past estimated: Jun 7, 2024 | INTERVENTIONAL
| Arrhythmias, Cardiac
Atrial Fibrillation
Atrial Flutter |
A Study to Test Whether BI 685509 Alone or in Combination With Empagliflozin Helps People With Liver Cirrhosis Caused by Viral Hepatitis or Non-alcoholic Steatohepatitis (NASH) Who Have High Blood Pressure in the Portal Vein (Main Vessel Going to the Liver)
Boehringer Ingelheim | Phase 2 Completed Jun 7, 2024 | INTERVENTIONAL
| Hypertension, Portal
Liver Diseases |
A Study of HSK31858 in Participants with Non-Cystic Fibrosis Bronchiectasis
Haisco Pharmaceutical Group Co., Ltd. | Phase 2 Completed Jun 7, 2024 | INTERVENTIONAL
| Non-cystic Fibrosis Bronchiectasis (NCFBE) |
ELU001 in Pediatric Subjects Who Have Relapsed and/or Refractory CBFA2T3-GLIS2-positive AML
Elucida Oncology | Phase 1 Completed Jun 7, 2024 | INTERVENTIONAL
| AML, Childhood
Acute Myeloid Leukemia
Refractory Pediatric AML
Relapsed Pediatric AML |
A Study to Evaluate the Effects of Renal Function on Pharmacokinetics and Safety of DA-8010
Dong-A ST Co., Ltd. | Phase 1 Completed Jun 7, 2024 | INTERVENTIONAL
| Healthy
Renal Impairment |
Prospective Clinical Evaluation of BD NRFit™ Devices and Accessories.
Becton, Dickinson and Company | Phase not applicable Completed Jun 7, 2024 | OBSERVATIONAL
| Analgesia
Anesthesia |
A Study to Evaluate the Safety and Immune Response of mRNA-1345, a Vaccine Targeting Respiratory Syncytial Virus (RSV), When Co-administered With a Fluzone HD, in Adults ≥65 Years of Age
ModernaTX, Inc. | Phase 3 Completed Jun 7, 2024 | INTERVENTIONAL
| Respiratory Syncytial Virus |
A Bioequivalence Study of Two Formulations of 10-mg Empagliflozin Tablets in Healthy Thai Volunteers Under Fasting Conditions
Pharma Nueva | Phase 1 Past estimated: Jun 7, 2024 | INTERVENTIONAL
| Healthy Vollunteer |
A Study to Evaluate the Drug Interaction Between Povorcitinib and the Oral Contraceptive Levonorgestrel/Ethinyl Estradiol in Healthy Adult Females
Incyte Corporation | Phase 1 Completed Jun 7, 2024 | INTERVENTIONAL
| Healthy Participants |
Perceived Wellbeing and Focus with Daily Greens Supplementation
Gainful | Phase not applicable Completed Jun 7, 2024 | INTERVENTIONAL
| Well-Being, Psychological |
Post Authorization Safety Profile of Booster IndoVac COVID-19 Vaccination
PT Bio Farma | Phase not applicable Completed Jun 7, 2024 | No interventions listed | COVID-19 |
A Phase II Study to Evaluate the Safety and Efficacy of SAL003 Combined With Atorvastatin in Hypercholesterolemia and Mixed Dyslipidemia
Shenzhen Salubris Pharmaceuticals Co., Ltd. | Phase 2 Completed Jun 7, 2024 | INTERVENTIONAL
| Hypercholesterolemia and Mixed Dyslipidemia |
Long-Term Extension Study With Eptinezumab as Preventive Treatment in Participants With Migraine in Japan
H. Lundbeck A/S | Phase 3 Completed Jun 8, 2024 | INTERVENTIONAL
| Migraine |
Altropane Dose for Imaging Patients With Suspected Parkinson's Disease
Fortrea | Phase 2 Completed Jun 8, 2024 | INTERVENTIONAL
| Movement Disorders
Parkinson Disease |
Altropane Dose for Imaging Patients With Suspected Parkinson's Disease
GE Healthcare | Phase 2 Completed Jun 8, 2024 | INTERVENTIONAL
| Movement Disorders
Parkinson Disease |
A Six-Month Prospective Follow-Up Study of WB001
Woebot Health | Phase not applicable Past estimated: Jun 8, 2024 | OBSERVATIONAL
| Postpartum Depression |
Screening for Neonatal Jaundice With a Mobile Health Device: a Validation Study in Oaxaca, Mexico.
Picterus AS | Phase not applicable Completed Jun 8, 2024 | INTERVENTIONAL
| Jaundice, Neonatal |
Evaluation of the Know Your OQ™ Oral Health Literacy Tool in an Indian Population
Colgate Palmolive | Phase not applicable Completed Jun 8, 2024 | No interventions listed | Oral Health Literacy |
Imaging-Guided Vessel Sizing in the Tibial Arteries
Abbott Medical Devices | Phase not applicable Completed Jun 9, 2024 | OBSERVATIONAL
| Critical Limb Ischemia
Peripheral Artery Disease |
Evaluate the Safety and Efficacy of Ferric Maltol Oral Suspension vs. Ferrous Sulfate Oral Liquid in Children and Adolescents Aged 2 to 17 Years With Iron-deficiency Anaemia, With a Single Arm Study in Infants Aged 1 Month to Less Than 2 Years
Shield Therapeutics | Phase 3 Completed Jun 9, 2024 | INTERVENTIONAL
| Anemia
Iron-deficiency |
Smart Textile Sensor System for Health Monitoring
Nanowear Inc. | Recruiting
Phase not applicable Past estimated: Jun 9, 2024 | OBSERVATIONAL
| Blood Pressure
Heart Disease Chronic
Heart Rhythm Disorder
Pulmonary Disease, Chronic Obstructive |
A Study of Single and Multiple Oral Doses of TollB-001
Toll Biotech Co. Ltd. (Beijing) | Recruiting
Phase 1 Past estimated: Jun 9, 2024 | INTERVENTIONAL
| Rheumatoid Arthritis |
A Study of Genolair in Two Dosage Forms (Solution and Lyophilisate) and Xolair® in Healthy Volunteers
AO GENERIUM | Phase 1 Completed Jun 9, 2024 | INTERVENTIONAL
| Allergic Asthma |
HORIZON: A Phase II Study to Evaluate the Safety and Efficacy of Two Doses of GT005
Gyroscope Therapeutics Limited | Phase 2 Completed Jun 10, 2024 | INTERVENTIONAL
| Dry Age-related Macular Degeneration |
HORIZON: A Phase II Study to Evaluate the Safety and Efficacy of Two Doses of GT005
Novartis Pharmaceuticals | Phase 2 Completed Jun 10, 2024 | INTERVENTIONAL
| Dry Age-related Macular Degeneration |
Ultra Curto (Ultra Short) TB Prevention Therapy
Sanofi | Phase 4 Completed Jun 10, 2024 | INTERVENTIONAL
| Tuberculosis |
Romosozumab in Women With Chronic SCI
Amgen | Phase 2 Completed Jun 10, 2024 | INTERVENTIONAL
| Bone Loss
Chronic Spinal Paralysis
Osteopenia, Osteoporosis
Osteoporosis
Spinal Cord Injuries |
Phase 1 Safety and Tolerability Study of MSK-DA01 Cell Therapy for Advanced Parkinson's Disease
BlueRock Therapeutics | Phase 1 Completed Jun 10, 2024 | INTERVENTIONAL
| Advanced Parkinson's Disease |
Bridge to HOPE: Hypothermic Oxygenated Perfusion Versus Cold Storage Prior to Liver Transplantation
Bridge to Life Ltd. | Phase not applicable Completed Jun 10, 2024 | INTERVENTIONAL
| End Stage Liver DIsease
Liver Failure |
A Study to Assess Change in Disease Activity and Adverse Events of Adalimumab in Chinese Participants Requiring High Dose Corticosteroids for Active Non-Infectious Intermediate, Posterior, or Pan-Uveitis
AbbVie | Phase 4 Completed Jun 10, 2024 | INTERVENTIONAL
| Non-infectious Intermediate Posterior- or Pan-uveitis |
Study to Evaluate the Safety, Tolerability, and PK of Pacritinib
CTI BioPharma | Phase 1 Completed Jun 10, 2024 | INTERVENTIONAL
| Hepatic Impairment |
Study to Evaluate the Safety, Tolerability, and PK of Pacritinib
PPD Development, LP | Phase 1 Completed Jun 10, 2024 | INTERVENTIONAL
| Hepatic Impairment |
A Study of Safety and Drug Levels of HIB210 in Healthy Volunteers
HI-Bio, A Biogen Company | Phase 1 Completed Jun 10, 2024 | INTERVENTIONAL
| Autoimmunity
Immune System Diseases |
Retrospective Medical Chart Review Study to Describe the Experience of SLE Patients Treated With Anifrolumab in the Early Access Programs.
AstraZeneca | Phase not applicable Completed Jun 10, 2024 | OBSERVATIONAL
| Systemic Lupus Erythematosus |
Cycloferon for Post-exposure Prophylaxis of Acute Respiratory Viral Infections and Influenza
POLYSAN Scientific & Technological Pharmaceutical Company | Phase 3 Completed Jun 10, 2024 | INTERVENTIONAL
| Acute Respiratory Infection
Influenza |
A Study to Test How Either a Capsule or a Tablet With NDec (Decitabine and Tetrahydrouridine) Works in the Body of Healthy People
Novo Nordisk A/S | Phase 1 Completed Jun 10, 2024 | INTERVENTIONAL
| Healthy Volunteers |
A Study to Learn About How BAY2927088 Affects the Level of Dabigatran or Rosuvastatin in the Blood When These Drugs Are Taken Together in Healthy Participants
Bayer | Phase 1 Completed Jun 10, 2024 | INTERVENTIONAL
| Advanced Non-small Cell Lung Cancer
EGFR Mutation
HER2 Mutation
Healthy Volunteers |
A Study to Evaluate the Pharmacokinetics and Safety Between HCP2303 and Co-administration of Each Component in Healthy Volunteers
Hanmi Pharmaceutical Company Limited | Phase 1 Completed Jun 10, 2024 | INTERVENTIONAL
| Healthy |
Tandem Freedom Feasibility Study 1
Tandem Diabetes Care, Inc. | Phase not applicable Completed Jun 10, 2024 | INTERVENTIONAL
| Diabetes Mellitus, Type 1 |
Lebrikizumab Pen Ease of Use in Participants With Atopic Dermatitis
Eli Lilly and Company | Phase 3 Completed Jun 10, 2024 | INTERVENTIONAL
| Atopic Dermatitis |
FLT3 Clonal Evolution in Patients With Acute Myeloid Leukemia
Astellas Pharma Inc | Phase not applicable Completed Jun 10, 2024 | No interventions listed | Acute Myeloid Leukemia |
Personalized Management of Autoimmune Diseases With AI-Guided (GenAIS TM) Dietary Supplementation
Triangel Scientific | Phase not applicable Completed Jun 10, 2024 | INTERVENTIONAL
| Autoimmune Diseases |
A Study to Learn About the Benefit of Rimegepant Versus Triptans in Migraine Buddy App Users.
Pfizer | Phase not applicable Completed Jun 10, 2024 | No interventions listed | Migraine |
Evaluating the Effect of a Supplement on Symptoms of Hormonal Imbalance.
Citruslabs | Phase not applicable Completed Jun 10, 2024 | INTERVENTIONAL
| Women's Health |
Evaluating the Effect of a Supplement on Symptoms of Hormonal Imbalance.
Perelel Inc. | Phase not applicable Completed Jun 10, 2024 | INTERVENTIONAL
| Women's Health |
Phase 1 Study in Diabetes Mellitus(Fed)
Huons Co., Ltd. | Phase 1 Completed Jun 10, 2024 | INTERVENTIONAL
| Diabete Mellitus
Diabetes |
Lactoferrin Alone ,Iron Alone Vs Lactoferrin Plus Iron for Treatment of Iron Deficiency Anemia
Egymedicalpedia | Phase not applicable Completed Jun 10, 2024 | INTERVENTIONAL
| Iron Deficiency Anemia |
Technical Validation of MR Biomarkers of Obesity-Associated NAFLD
GE Healthcare | Phase not applicable Completed Jun 11, 2024 | OBSERVATIONAL
| Non-Alcoholic Fatty Liver Disease
Non-alcoholic Steatohepatitis |
Technical Validation of MR Biomarkers of Obesity-Associated NAFLD
Pfizer | Phase not applicable Completed Jun 11, 2024 | OBSERVATIONAL
| Non-Alcoholic Fatty Liver Disease
Non-alcoholic Steatohepatitis |
Technical Validation of MR Biomarkers of Obesity-Associated NAFLD
Siemens Medical Solutions | Phase not applicable Completed Jun 11, 2024 | OBSERVATIONAL
| Non-Alcoholic Fatty Liver Disease
Non-alcoholic Steatohepatitis |
Radiotherapy, Carboplatin/Paclitaxel and Nivolumab for High Risk HPV-related Head and Neck Cancer
Bristol-Myers Squibb | Phase 2 Completed Jun 11, 2024 | INTERVENTIONAL
| Oropharynx Squamous Cell Carcinoma |
Evaluation of Efficacy and Safety of Neoadjuvant Treatment With Pamrevlumab in Combination With Chemotherapy (Either Gemcitabine Plus Nab-paclitaxel or FOLFIRINOX) in Participants With Locally Advanced, Unresectable Pancreatic Cancer
FibroGen | Phase 3 Completed Jun 11, 2024 | INTERVENTIONAL
| Pancreatic Cancer Non-resectable |
A Study Evaluating Oral Atogepant for the Prevention of Migraine in Japanese Participants With Chronic or Episodic Migraine
AbbVie | Phase 3 Completed Jun 11, 2024 | INTERVENTIONAL
| Chronic Migraine
Episodic Migraine |
Oral ISL QM as PrEP in Cisgender Women at High Risk for HIV-1 Infection (MK-8591-022)
Merck Sharp & Dohme LLC | Phase 3 Completed Jun 11, 2024 | INTERVENTIONAL
| HIV-I
Human Immunodeficiency Virus Type 1
Prophylaxis |
Oral Nutritional Supplementation in Children at Risk of Undernutrition
Abbott Nutrition | Phase not applicable Completed Jun 11, 2024 | INTERVENTIONAL
| Undernutrition |
BIO|CONCEPT.Amvia Study
Biotronik Australia Pty Ltd. | Phase not applicable Completed Jun 11, 2024 | INTERVENTIONAL
| Bradycardia
Heart Failure |
BIO|CONCEPT.Amvia Study
Biotronik SE & Co. KG | Phase not applicable Completed Jun 11, 2024 | INTERVENTIONAL
| Bradycardia
Heart Failure |
Impact on the Use of Antibiotics of a Multimodal Algorithm for the Diagnosis and Management of Acute Community-acquired Pneumonia in the Emergency Room
QIAGEN Gaithersburg, Inc | Phase not applicable Completed Jun 11, 2024 | INTERVENTIONAL
| Community-acquired Pneumonia |
To Compare the Efficacy and Safety of INS068 and Insulin Glargine in Subjects With Type 2 Diabetes Mellitus Treated With Basal Insulin.
Jiangsu HengRui Medicine Co., Ltd. | Phase 3 Completed Jun 11, 2024 | INTERVENTIONAL
| Type 2 Diabetes Mellitus |
Studying the Safety, Efficacy, and Pharmacokinetic Characteristics of BNCT in Patients With Recurrent High-grade Gliomas
Dawonmedax Co., Ltd. | Phase 1 / Phase 2 Completed Jun 11, 2024 | INTERVENTIONAL
| Recurrent Anaplastic Astrocytoma
Recurrent Anaplastic Oligodendroglioma
Recurrent Glioblastoma
Recurrent High-grade Glioma |
Assessment of Left Ventricular Filling Pressure by Applying Artificial Intelligence Algorithms to Left Atrial Speckle-tracking Echocardiography
Philips Healthcare | Phase not applicable Past estimated: Jun 11, 2024 | OBSERVATIONAL
| Echocardiography
Heart Failure, Diastolic |
Evaluation of the F&P Caramel Nasal Mask, US, 2023
Fisher and Paykel Healthcare | Phase not applicable Completed Jun 11, 2024 | INTERVENTIONAL
| Obstructive Sleep Apnea |
Compare the Pharmacokinetics and Safety of BAT3306 Injection Versus KEYTRUDA® Administered in Healthy Male Subjects
Bio-Thera Solutions | Phase 1 Completed Jun 11, 2024 | INTERVENTIONAL
| Healthy Male Subjects |
Ovulation Triggering 2 vs 3 Follicles
Clinique Ovo | Phase not applicable Completed Jun 11, 2024 | OBSERVATIONAL
| Infertility |
A Study to Assess the Pharmacokinetics and Safety of Budesonide, Glycopyrronium, & Formoterol (BGF) Metered Dose Inhaler (MDI) With a Next-Generation Propellant (NGP) With a Spacer, BGF MDI Hydrofluoroalkane (HFA) With a Spacer, as Well as BGF MDI NGP Without a Spacer
AstraZeneca | Phase 1 Completed Jun 11, 2024 | INTERVENTIONAL
| Chronic Obstructive Pulmonary Disease |
A Study to Assess the Pharmacokinetics and Safety of Budesonide, Glycopyrronium, & Formoterol (BGF) Metered Dose Inhaler (MDI) With a Next-Generation Propellant (NGP) With a Spacer, BGF MDI Hydrofluoroalkane (HFA) With a Spacer, as Well as BGF MDI NGP Without a Spacer
Parexel | Phase 1 Completed Jun 11, 2024 | INTERVENTIONAL
| Chronic Obstructive Pulmonary Disease |
A Study to Learn About How Food and Esomeprazole Affect the Level of BAY2927088 in the Blood When Taken Together in Healthy Participants
Bayer | Phase 1 Completed Jun 11, 2024 | INTERVENTIONAL
| Advanced Non-small Cell Lung Cancer
EGFR Mutation
HER2 Mutation
Healthy Volunteers |
A Clinical Trial to Assess the Efficacy and Safety of Subjects With Dry Eye Disease
Aldeyra Therapeutics, Inc. | Phase 3 Completed Jun 11, 2024 | INTERVENTIONAL
| Dry Eye Disease |
Comparison of Complarate® (Tocilizumab Biosimilar) and Actemra® in Patients With Rheumatoid Arthritis
AO GENERIUM | Phase 3 Completed Jun 11, 2024 | INTERVENTIONAL
| Rheumatoid Arthritis |
A Study of Long-term Effects of Vedolizumab Subcutaneous in Adults With Ulcerative Colitis and Crohn's Disease
Takeda | Phase 3 Completed Jun 12, 2024 | INTERVENTIONAL
| Colitis, Ulcerative
Crohn's Disease |
GOLDEN STUDY: A Study to Assess Safety and Efficacy of Multiple Doses of IONIS-FB-LRx in Participants With Geographic Atrophy Secondary to Age-Related Macular Degeneration (AMD)
Ionis Pharmaceuticals, Inc. | Phase 2 Completed Jun 12, 2024 | INTERVENTIONAL
| Geographic Atrophy
Macular Degeneration |
Cryopreserved Human Umbilical Cord (TTAX01) for Late Stage, Complex Non-healing Diabetic Foot Ulcers (AMBULATE DFU)
BioTissue Holdings, Inc | Phase 3 Completed Jun 12, 2024 | INTERVENTIONAL
| Diabetic Foot Infection
Non-healing Diabetic Foot Ulcer
Non-healing Wound |
LUMINOS-103: A Basket Trial of Safety & Efficacy of Lerapolturev With or Without Checkpoint Inhibitors
Istari Oncology, Inc. | Phase 1 / Phase 2 Completed Jun 12, 2024 | INTERVENTIONAL
| Bladder Cancer
Non-muscle-invasive Bladder Cancer
Solid Tumor |
Study of TG-1801 Alone or in Combination With Ublituximab in Subjects With B-Cell Lymphoma or Chronic Lymphocytic Leukemia
TG Therapeutics, Inc. | Phase 1 Completed Jun 12, 2024 | INTERVENTIONAL
| Aggressive Lymphoma
CLL
DLBCL
Follicular Lymphoma
Indolent Lymphoma
MCL
Marginal Zone Lymphoma
Mediastinal Large B-cell Lymphoma
Richter's Transformation
SLL |
StrataPATH™ (Precision Indications for Approved Therapies)
Gilead Sciences | Phase 2 Completed Jun 12, 2024 | INTERVENTIONAL
| Advanced Solid Tumor
Cancer |
StrataPATH™ (Precision Indications for Approved Therapies)
Merck Sharp & Dohme LLC | Phase 2 Completed Jun 12, 2024 | INTERVENTIONAL
| Advanced Solid Tumor
Cancer |
StrataPATH™ (Precision Indications for Approved Therapies)
Pfizer | Phase 2 Completed Jun 12, 2024 | INTERVENTIONAL
| Advanced Solid Tumor
Cancer |
StrataPATH™ (Precision Indications for Approved Therapies)
Strata Oncology | Phase 2 Completed Jun 12, 2024 | INTERVENTIONAL
| Advanced Solid Tumor
Cancer |
A Study to Test Whether Two Different Doses of Avenciguat Help People With Liver Cirrhosis and High Blood Pressure in the Portal Vein (Main Vessel Going to the Liver)
Boehringer Ingelheim | Phase 2 Completed Jun 12, 2024 | INTERVENTIONAL
| Hypertension, Portal |
A Study of Neoantigen mRNA Personalised Cancer in Patients With Advanced Solid Tumors
Stemirna Therapeutics | Recruiting
Phase 1 Past estimated: Jun 12, 2024 | INTERVENTIONAL
| Solid Tumor |
Comparison of Efficacy, Pharmacodynamics, Safety, and Immunogenicity Between Bmab 1000 and Prolia® in Postmenopausal Women With Osteoporosis
Biocon Biologics UK Ltd | Phase 3 Completed Jun 12, 2024 | INTERVENTIONAL
| Postmenopausal Women With Osteoporosis |
Characterizing the Intraocular Scattering at Different Wavelengths
Johnson & Johnson Vision Care, Inc. | Phase not applicable Completed Jun 12, 2024 | INTERVENTIONAL
| Vision, Ocular |
Evuzamitide in PET/CT to Measure Potential Therapeutic Response in ATTR
Attralus, Inc. | Phase 2 Completed Jun 12, 2024 | INTERVENTIONAL
| Transthyretin Amyloidosis |
Clinical Evaluation of a Myopia Control Lens in Slowing Myopia Progression (CSL)
Essilor International | Phase not applicable Completed Jun 12, 2024 | INTERVENTIONAL
| Myopia |
A Study to Evaluate Pharmacokinetics (PK) and Safety of Inaxaplin in Participants With Renal Impairment
Vertex Pharmaceuticals Incorporated | Phase 1 Completed Jun 12, 2024 | INTERVENTIONAL
| Renal Impairment |
A Study to Test How Kidney Problems Influence the Blood Concentrations of Efgartigimod
argenx | Phase 1 Completed Jun 12, 2024 | INTERVENTIONAL
| Renal Impairment |
Crome/Cobalt Respiration Study
Medtronic Cardiac Rhythm and Heart Failure | Phase not applicable Completed Jun 12, 2024 | OBSERVATIONAL
| Heart Failure |
VH4524184 Proof-of-Concept in Treatment-Naïve Adults Living With HIV-1
GlaxoSmithKline | Phase 2 Completed Jun 12, 2024 | INTERVENTIONAL
| HIV Infections |
VH4524184 Proof-of-Concept in Treatment-Naïve Adults Living With HIV-1
ViiV Healthcare | Phase 2 Completed Jun 12, 2024 | INTERVENTIONAL
| HIV Infections |
Glin3: Assessment of the Glycemic Responses to Nutritional Products
Nutricia Research | Phase not applicable Completed Jun 12, 2024 | INTERVENTIONAL
| Glycemic Response |
A Phase 1 Study Evaluating the Safety, Pharmacokinetics, and Pharmacodynamics, of VX-993
Vertex Pharmaceuticals Incorporated | Phase 1 Completed Jun 12, 2024 | INTERVENTIONAL
| Pain |
Sitagliptin and Metformin Hydrochloride Tablets 50 mg/1000 mg Relative to Originator
Bio-innova Co., Ltd | Phase 1 Past estimated: Jun 12, 2024 | INTERVENTIONAL
| Healthy Subjects |
ETNA-MS Device Validation Study
Innodem Neurosciences | Phase not applicable Past estimated: Jun 12, 2024 | OBSERVATIONAL
| Multiple Sclerosis |
Pharmacokinetics of HD-P023 and Co-administration of Teneligliptin and Empagliflozin High in Healthy Volunteers
Handok Inc. | Phase 1 Completed Jun 12, 2024 | INTERVENTIONAL
| Healthy Volunteers |
A Study to Learn About How Itraconazole and Carbamazepine Affect the Level of BAY2927088 in the Blood When These Drugs Are Taken Together by Healthy Participants
Bayer | Phase 1 Completed Jun 12, 2024 | INTERVENTIONAL
| Advanced Non-small Cell Lung Cancer
EGFR Mutation
HER2 Mutation
Healthy Volunteers |
Observational Cohort Study Evaluating the Effectiveness of an Educational Program (NGAYDAUTIEN) in Combination With Perindopril and Amlodipine Single-Pill Combination in Hypertensive Patients naïve of Antihypertensive Treatments.
Servier Affaires Médicales | Phase not applicable Completed Jun 12, 2024 | No interventions listed | Hypertension |
Secur-Fit Advanced Outcomes Study
Stryker Orthopaedics | Phase not applicable Completed Jun 13, 2024 | INTERVENTIONAL
| Arthroplasty, Replacement, Hip |
A Research Study of How Well Macimorelin Works to Find Out if Children Have a Lack of Growth Hormone and How Safe it is
AEterna Zentaris | Phase 3 Completed Jun 13, 2024 | INTERVENTIONAL
| Growth Hormone Deficiency |
Duvelisib Plus Docetaxel In Recurrent/Metastatic HNSCC
Secura Bio, Inc. | Phase 2 Completed Jun 13, 2024 | INTERVENTIONAL
| Advanced Head and Neck Cancer
Advanced Head and Neck Squamous Cell Carcinoma
Metastatic Head and Neck Cancer
Recurrent Squamous Cell Carcinoma of the Head and Neck
Squamous Cell Carcinoma of the Head and Neck (SCCHN) |
A Study on the Immune Response and Safety of Various Potencies of an Investigational Chickenpox Vaccine Compared With a Marketed Chickenpox Vaccine, Given to Healthy Children 12 to 15 Months of Age
GlaxoSmithKline | Phase 2 Completed Jun 13, 2024 | INTERVENTIONAL
| Chickenpox |
Trilaciclib in Patients Receiving Sacituzumab Govitecan-hziy for Triple Negative Breast Cancer
G1 Therapeutics, Inc. | Phase 2 Completed Jun 13, 2024 | INTERVENTIONAL
| Triple Negative Breast Cancer |
A Study of SHR-A1909 in Subjects With Advanced Malignant Tumors
Shanghai Hengrui Pharmaceutical Co., Ltd. | Phase 1 Completed Jun 13, 2024 | INTERVENTIONAL
| Advanced Malignant Cancer |
Study of Interest of Stiripentol and Carbamazepine in the Treatment of Patients With Pharmacoresistant Focal Epilepsies
Biocodex | Phase 4 Completed Jun 13, 2024 | INTERVENTIONAL
| Pharmacoresistant Focal Epilepsies |
A Randomized, Double-Blind, Placebo-Controlled Study With an Open-Label Period on Efficacy and Safety of Fremanezumab in Chinese Adults With Migraine
Teva Branded Pharmaceutical Products R&D, Inc. | Phase 3 Completed Jun 13, 2024 | INTERVENTIONAL
| Migraine |
A Research Study Looking at the Safety of Multiple Doses of ZP8396 and How it Works in the Body of Healthy Participants
Zealand Pharma | Phase 1 Completed Jun 13, 2024 | INTERVENTIONAL
| Healthy Volunteers
Obese
Overweight |
Phenotyping Responses to Systemic Corticosteroids in the Management of Asthma Attacks
BioMérieux | Phase not applicable Completed Jun 13, 2024 | OBSERVATIONAL
| Asthma |
Visual Performance of the Vivity IOL in Post-myopic LASIK and PRK Patients
Sengi | Phase not applicable Completed Jun 13, 2024 | OBSERVATIONAL
| Cataract |
Investigating Mild Cognitive Impairment in Patients And Controls With TD-fNIRS
Kernel | Phase not applicable Completed Jun 13, 2024 | OBSERVATIONAL
| Mild Cognitive Impairment |
Phase I Clinical Study of ZL-82 Tablets
Chengdu Zenitar Biomedical Technology Co., Ltd | Phase 1 Completed Jun 13, 2024 | INTERVENTIONAL
| Inflammatory Bowel Disease - IBD1
Rheumatoid Arthritis (RA) |
A Study to Assess the Relative Bioavailability of 5 mg Mavacamten Opened Capsule Administered Via Nasogastric Tube Compared to Intact Oral 5 mg Mavacamten in Healthy Participants
Bristol-Myers Squibb | Phase 1 Completed Jun 13, 2024 | INTERVENTIONAL
| Healthy Volunteers |
A Clinical Trial to Evaluate the Food Effect of CKD-378
Chong Kun Dang Pharmaceutical | Phase 1 Completed Jun 13, 2024 | INTERVENTIONAL
| Type II Diabetes Mellitus |
Accuracy of the EvieMED Ring Pulse Oximeter in Profound Hypoxia
Movano Health | Phase not applicable Past estimated: Jun 13, 2024 | OBSERVATIONAL
| Hypoxia |
Multicenter, Prospective Study to Determine the Performance and Safety of the New M-HA30 Architect Device on the Quality of the Skin: HEBE8 Study
Laboratoires FILLMED | Phase not applicable Completed Jun 13, 2024 | INTERVENTIONAL
| Skin Aging |
Feasibility of the LUM Imaging System for Detection of Cancer to the Brain
Lumicell, Inc. | Phase not applicable Completed Jun 14, 2024 | INTERVENTIONAL
| Glioblastoma
Low Grade Glioma of Brain
Metastasis to Brain |
PROactive Evaluation of Function to Avoid CardioToxicity
Myocardial Solutions | Phase 2 Completed Jun 14, 2024 | INTERVENTIONAL
| Breast Cancer
Cardiotoxicity
Leukemia
Lung Cancer
Lymphoma
Myeloma
Sarcoma |
Tazemetostat in Combination With Doxorubicin as Frontline Therapy for Advanced Epithelioid Sarcoma
Epizyme, Inc. | Phase 3 Completed Jun 14, 2024 | INTERVENTIONAL
| Advanced Epithelioid Sarcoma
Advanced Soft-tissue Sarcoma |
OLE of Phase 2b/3 Study ANAVEX2-73-AD-004
Anavex Australia Pty Ltd. | Phase 2 / Phase 3 Completed Jun 14, 2024 | INTERVENTIONAL
| Alzheimer Disease |
OLE of Phase 2b/3 Study ANAVEX2-73-AD-004
Anavex Germany GmbH | Phase 2 / Phase 3 Completed Jun 14, 2024 | INTERVENTIONAL
| Alzheimer Disease |
OLE of Phase 2b/3 Study ANAVEX2-73-AD-004
Anavex Life Sciences Corp. | Phase 2 / Phase 3 Completed Jun 14, 2024 | INTERVENTIONAL
| Alzheimer Disease |
Study to Evaluate the Efficacy (Effect on Disease) and Safety of Finerenone in Participants With Heart Failure and Left Ventricular Ejection Fraction (Proportion of Blood Expelled Per Heart Stroke) Greater or Equal to 40%
Bayer | Phase 3 Completed Jun 14, 2024 | INTERVENTIONAL
| Heart Failure |
A Study to Evaluate the Safety, Reactogenicity, and Effectiveness of mRNA-1273 Vaccine in Adolescents 12 to <18 Years Old to Prevent COVID-19
ModernaTX, Inc. | Phase 2 / Phase 3 Completed Jun 14, 2024 | INTERVENTIONAL
| SARS-CoV-2 |
A Trial Monitoring Therapy Pathways of Asthma Patients Treated With an Extrafine ICS/LABA/LAMA Single-inhaler Triple Therapy in a Real-world Setting and Characterizing the Effects on Health-related Out-comes
Chiesi UK | Phase not applicable Completed Jun 14, 2024 | OBSERVATIONAL
| Asthma |
A Trial Monitoring Therapy Pathways of Asthma Patients Treated With an Extrafine ICS/LABA/LAMA Single-inhaler Triple Therapy in a Real-world Setting and Characterizing the Effects on Health-related Out-comes
Gesellschaft für Therapieforschung mbH | Phase not applicable Completed Jun 14, 2024 | OBSERVATIONAL
| Asthma |
A Study to Evaluate the Safety and Effectiveness of a Digital Therapeutic
Oui Therapeutics, Inc. | Phase 2 / Phase 3 Completed Jun 14, 2024 | INTERVENTIONAL
| Suicidal Ideation
Suicide, Attempted |
A Study to Assess the Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of AZD0780 in Healthy Subjects
AstraZeneca | Phase 1 Completed Jun 14, 2024 | INTERVENTIONAL
| Dyslipidemia |
A Study to Assess the Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of AZD0780 in Healthy Subjects
Parexel | Phase 1 Completed Jun 14, 2024 | INTERVENTIONAL
| Dyslipidemia |
A Study to Evaluate D-1553 in Combination Therapy in Non-Small Cell Lung Cancer
InventisBio Co., Ltd | Phase 1 / Phase 2 Completed Jun 14, 2024 | INTERVENTIONAL
| NSCLC |
Evaluation of Quality of Vision and Visual Outcomes With Bilateral Implantation of the Clareon PanOptix Intraocular Lens
Sengi | Phase not applicable Completed Jun 14, 2024 | OBSERVATIONAL
| Cataract |
Effectiveness and Safety of Restylane Lyft Lidocaine for Jawline Definition
Galderma R&D | Phase not applicable Completed Jun 14, 2024 | INTERVENTIONAL
| Jawline Definition |
Immuno-positron Emission Tomography Study of 89Zr-S095012 in Patients With Advanced Solid Tumours
ADIR, a Servier Group company | Phase 1 Completed Jun 14, 2024 | INTERVENTIONAL
| Advanced Solid Tumor |
A Study in Male and Female Adult Participants With Chronic Rhinosinusitis With Nasal Polyps In the Greater Gulf Region
Sanofi | Phase not applicable Completed Jun 14, 2024 | No interventions listed | Chronic Rhinosinusitis With Nasal Polyps |
A Study in Healthy Men to Test How Well Multiple Doses of BI 1839100 Are Tolerated and How BI 1839100 Influences the Amount of Other Medicines in the Blood
Boehringer Ingelheim | Phase 1 Completed Jun 14, 2024 | INTERVENTIONAL
| Healthy |
Single and Multiple Ascending Dose Study to Assess Safety, Tolerability and Pharmacokinetics of NM-101
Neuramedy Co. Ltd. | Not yet recruiting
Phase 1 Past estimated: Jun 14, 2024 | INTERVENTIONAL
| Parkinson Disease |
Single and Multiple Ascending Dose Study to Assess Safety, Tolerability and Pharmacokinetics of NM-101
Quotient Sciences | Not yet recruiting
Phase 1 Past estimated: Jun 14, 2024 | INTERVENTIONAL
| Parkinson Disease |
The Efficacy and Safety of Protoxin in Subjects With Moderate to Severe Glabellar Lines
Protox Inc. | Phase 3 Completed Jun 14, 2024 | INTERVENTIONAL
| Glabellar Frown Lines |
Pharmacokinetics and Pharmacodynamics of Subcutaneous vs Intravenous Furosemide in Healthy Volunteers
scPharmaceuticals, Inc. | Phase 1 Completed Jun 14, 2024 | INTERVENTIONAL
| Fluid Overload
Heart Failure |
UV1 Vaccine With Pembrolizumab for Patients With Unresectable or Metastatic Melanoma
Ultimovacs ASA | Phase 1 Completed Jun 15, 2024 | INTERVENTIONAL
| Malignant Melanoma |
EmboTrap ® II Revascularization Device (Neuravi) in Acute Ischemic Stroke
Neuravi Limited | Recruiting
Phase not applicable Past estimated: Jun 15, 2024 | OBSERVATIONAL
| Acute Ischemic Stroke |
Radiotherapy in IDH Mutated Glioma: Evaluation of Late Outcomes
HollandPTC | Phase not applicable Completed Jun 15, 2024 | No interventions listed | Astrocytoma, Grade II
Astrocytoma, Grade III
Oligodendroglioma
Oligodendroglioma, Anaplastic |
Ticagrelor With and Without Aspirin in Patients With Diabetes Mellitus
AstraZeneca | Phase 4 Completed Jun 15, 2024 | INTERVENTIONAL
| Coronary Artery Disease
Diabetes Mellitus, Type 2 |
Motivating Occupational Virtual Experiences In Therapy for Kids
Barron Associates, Inc. | Phase not applicable Completed Jun 15, 2024 | INTERVENTIONAL
| Occupational Therapy
Virtual Reality |
In Vitro Analysis of Human Urine With the CloudCath Device
CloudCath | Phase not applicable Past estimated: Jun 15, 2024 | OBSERVATIONAL
| Urinary Tract Infections |
Management of Moderate POstoperative Recurrence in Crohn's Disease: a randoMizEd contROLled Trial of Therapeutic Escalation, the POMEROL Trial. (POMEROL)
Celltrion | Recruiting
Phase 4 Past estimated: Jun 15, 2024 | INTERVENTIONAL
| Crohn Disease |
Sacral Fracture Fusion/Fixation for Rapid Rehabilitation
SI-BONE, Inc. | Phase not applicable Completed Jun 15, 2024 | INTERVENTIONAL
| Fracture;Pelvis
Fragility Fracture
Insufficiency Fractures
Osteoporosis
Sacral Fracture
Sacroiliac; Fusion |
Study of AT-527 in Subjects With Normal and Impaired Renal Function
Atea Pharmaceuticals, Inc. | Phase 1 Completed Jun 15, 2024 | INTERVENTIONAL
| Healthy Volunteer
Renal Impairment |
Evaluation of CG-100 Intraluminal Bypass Device
Colospan Ltd. | Phase not applicable Past estimated: Jun 15, 2024 | INTERVENTIONAL
| Colorectal Cancer |
A Phase I Study of BC3402 as a Single Agent in Patients With MDS and CMML
Biocity Biopharmaceutics Co., Ltd. | Recruiting
Phase 1 Past estimated: Jun 15, 2024 | INTERVENTIONAL
| Hematologic Malignancy |
Selective Lipopolysaccharide Hemosorption in Maternal Sepsis
Efferon JSC | Phase not applicable Completed Jun 15, 2024 | INTERVENTIONAL
| Maternal sepsIs |
An Observational Study to Learn More How Chronic Kidney Disease Gradually Changes Over Time in Adults Using Electronic Healthcare Records (CKD Natural History Study)
Bayer | Phase not applicable Completed Jun 15, 2024 | OBSERVATIONAL
| Chronic Kidney Disease |
Safety and Tolerance of Increased Doses of SHR-2106 Injection in Healthy Subjects
Guangdong Hengrui Pharmaceutical Co., Ltd | Enrolling by invitation
Phase 1 Past estimated: Jun 15, 2024 | INTERVENTIONAL
| Kidney Transplantation |
Effect of an Exploratory Vehicle on Meibomian Gland Dysfunction in Patients With Demodex
Tarsus Pharmaceuticals, Inc. | Phase 2 Past estimated: Jun 15, 2024 | INTERVENTIONAL
| Blepharitis
Demodectic Blepharitis
Demodex Infestation of Eyelid
Meibomian Gland Dysfunction |
A Randomized-Controlled Trial to Examine the Effects of a Daily Probiotic Supplement on Common Symptoms of Menopause
Citruslabs | Phase not applicable Completed Jun 15, 2024 | INTERVENTIONAL
| Menopause |
A Randomized-Controlled Trial to Examine the Effects of a Daily Probiotic Supplement on Common Symptoms of Menopause
Maneuver Marketing | Phase not applicable Completed Jun 15, 2024 | INTERVENTIONAL
| Menopause |
A Multi-center, Early Feasibility Study of Using the Mechanical Tissue Resuscitation™ (MTR™) Therapy System for Removal of Excess Fluid in Subjects Who Have Had a Portion of Their Skull Removed to Expose the Dura/Brain and Require Drainage for Fluid Removal.
Renovo Concepts, Inc. | Phase not applicable Past estimated: Jun 15, 2024 | INTERVENTIONAL
| Clinical Need for Craniotomy or Craniectomy |
The Effect of the Probiotic Strains on Bone Mineral Density in Postmenopausal Women
Nordic Biotic Sp. z o.o. | Phase not applicable Past estimated: Jun 15, 2024 | INTERVENTIONAL
| Bone Density, Low
Osteoporosis, Postmenopausal |
A Multi-site Clinical Evaluation of Capillary Blood Samples Collected Using the Tasso+SST Device for Downstream Analyte Testing
Tasso Inc. | Phase not applicable Past estimated: Jun 15, 2024 | OBSERVATIONAL
| Serum Analytes |
Bioavailability and Food Effects of HRS-7535 Tablets With Different Formulating Processes in Healthy Subjects
Shandong Suncadia Medicine Co., Ltd. | Phase 1 Past estimated: Jun 15, 2024 | INTERVENTIONAL
| Type 2 Diabetes Mellitus |
Comparative Study of Two Hyaluronic Acid Formulation in the Management of Knee Osteoarthritis
Fidia Farmaceutici s.p.a. | Phase not applicable Completed Jun 15, 2024 | INTERVENTIONAL
| Osteoarthritis, Knee |
Breathe-Easy: Hayfever Care for Patients With Seasonal Allergic Rhinitis
Innowage Limited | Phase not applicable Completed Jun 15, 2024 | INTERVENTIONAL
| Allergic Rhinitis
Allergy Pollen
Hayfever |
Study to Evaluate the Pharmacokinetics, Safety and Tolerability of Recifercept in Healthy Chinese Participants
Pfizer | Phase 1 Past estimated: Jun 16, 2024 | INTERVENTIONAL
| Healthy |
Impact of BacT/Alert® VIRTUO®, BioFire® Blood Culture Identification 2-BCID2 and REVEAL® (bioMérieux) on the Optimization of Antibiotic Therapy for Gram-negative Bacteremia in the ICU
BioMérieux | Phase not applicable Completed Jun 16, 2024 | OBSERVATIONAL
| Bacteremia |
A Trial to Investigate the Single Dose Pharmacokinetics of Insulin Degludec/Liraglutide in Healthy Chinese Subjects
Tonghua Dongbao Pharmaceutical Co.,Ltd | Phase 1 Past estimated: Jun 16, 2024 | INTERVENTIONAL
| Healthy |
A Study of LGK974 in Patients With Malignancies Dependent on Wnt Ligands
Novartis Pharmaceuticals | Phase 1 Completed Jun 17, 2024 | INTERVENTIONAL
| BRAF Mutant Colorectal Cancer
Cervical Squamous Cell Cancer
Esophageal Squamous Cell Cancer
Head and Neck Squamous Cell Cancer
Lung Squamous Cell Cancer
Melanoma
Pancreatic Cancer
Triple Negative Breast Cancer |
Multi-Arm Study to Test the Efficacy of Immunotherapeutic Agents in Multiple Sarcoma Subtypes
MedImmune LLC | Phase 2 Completed Jun 17, 2024 | INTERVENTIONAL
| Advanced and/or Metastatic Sarcoma |
The Late Presenter Treatment Optimisation Study
Gilead Sciences | Phase 3 Completed Jun 17, 2024 | INTERVENTIONAL
| HIV/AIDS |
The Late Presenter Treatment Optimisation Study
Janssen Pharmaceuticals | Phase 3 Completed Jun 17, 2024 | INTERVENTIONAL
| HIV/AIDS |
Randomized Placebo Controlled Trial Evaluating the Efficacy of Pimavanserin, a Selective Serotonin 5-HydroxyTryptamine-2A (5HT2A) Inverse Agonist, to Treat Impulse Control Disorders in Parkinson's Disease.
ACADIA Pharmaceuticals Inc. | Phase 2 Completed Jun 17, 2024 | INTERVENTIONAL
| Parkinson Disease |
Hepcidin Mimetic in Patients With Polycythemia Vera (REVIVE)
Protagonist Therapeutics, Inc. | Phase 2 Completed Jun 17, 2024 | INTERVENTIONAL
| Polycythemia Vera |
Study to Evaluate Safety and Clinical Activity of AB122 in Biomarker Selected Participants With Advanced Solid Tumors
Arcus Biosciences, Inc. | Phase 1 Completed Jun 17, 2024 | INTERVENTIONAL
| Advanced Solid Tumors |
Study to Evaluate Safety and Clinical Activity of AB122 in Biomarker Selected Participants With Advanced Solid Tumors
Gilead Sciences | Phase 1 Completed Jun 17, 2024 | INTERVENTIONAL
| Advanced Solid Tumors |
Study to Evaluate Safety and Clinical Activity of AB122 in Biomarker Selected Participants With Advanced Solid Tumors
Strata Oncology | Phase 1 Completed Jun 17, 2024 | INTERVENTIONAL
| Advanced Solid Tumors |
Long Term Arrhythmia Risk and Cardiovascular Events in Hematopoietic Stem Cell Transplant
Medtronic | Phase not applicable Completed Jun 17, 2024 | OBSERVATIONAL
| Atrial Fibrillation
Atrial Flutter
Hematopoietic Stem Cell Transplant |
The Pharmacokinetic and Pharmacodynamic Effects of Oral Cannabidiol (CBD) Under Acute and Chronic Exposure Conditions
Canopy Growth Corporation | Phase 1 Completed Jun 17, 2024 | INTERVENTIONAL
| Cannabis, Drug Effects |
Device to Assist With Abdominal Access During Laparoscopic Surgery
TauTona Group | Phase not applicable Completed Jun 17, 2024 | INTERVENTIONAL
| Laparoscopic Surgery |
PiXL as a Treatment for Low Grade Myopia
Glaukos Corporation | Phase not applicable Past estimated: Jun 17, 2024 | INTERVENTIONAL
| Myopia
Refractive Errors |
Safety and Efficacy of Hyperbaric Oxygen Therapy for Long COVID Syndrome
Karolinska Trial Alliance | Phase 2 Completed Jun 17, 2024 | INTERVENTIONAL
| COVID-19
Post COVID Condition
Post COVID-19 Condition
Post COVID-19 Condition, Unspecified
Post-Acute COVID-19 Syndrome
Post-COVID Syndrome |
Evaluating the Safety, Tolerability, and Pharmacokinetics of BAT1006
Bio-Thera Solutions | Phase 1 Completed Jun 17, 2024 | INTERVENTIONAL
| Advanced Solid Tumor |
A Study of ABM-1310 in Patients With BRAF V600-Mutant Advanced Solid Tumors
ABM Therapeutics Shanghai Company Limited | Phase 1 Completed Jun 17, 2024 | INTERVENTIONAL
| Advanced Solid Tumor
BRAF V600 Mutation |
A Study to Investigate the Safety, Tolerability, and Pharmacokinetics of Monoclonal Antibody (mAb) in Patients With IPF (SAD).
Chiesi Farmaceutici S.p.A. | Phase 1 Completed Jun 17, 2024 | INTERVENTIONAL
| Idiopathic Pulmonary Fibrosis |
A Study For Treatment Of Paroxysmal Atrial Fibrillation (PAF) With The THERMOCOOL SMARTTOUCH SF Catheter and TRUPULSE Generator
Biosense Webster, Inc. | Phase not applicable Completed Jun 17, 2024 | INTERVENTIONAL
| Atrial Fibrillation |
An Observational Study of Moderna COVID-19 Bivalent Vaccines (Original and Omicron BA.4/BA.5) and 2023 Updated mRNA COVID-19 Vaccines (XBB.1.5)
ModernaTX, Inc. | Phase not applicable Completed Jun 17, 2024 | OBSERVATIONAL
| COVID-19 |
Consistently Assess Signs and Symptom of Heart Failure
Abbott | Recruiting
Phase not applicable Past estimated: Jun 17, 2024 | INTERVENTIONAL
| Heart Failure
Heart Failure With Preserved Ejection Fraction
Heart Failure With Reduced Ejection Fraction |
Mindfulness-based Cognitive Coping for ADRD Caregivers
Photozig, Inc. | Phase 2 Completed Jun 17, 2024 | INTERVENTIONAL
| Caregiver Stress |
The Prevalence Evaluation of Systemic Lupus Erythematosus in Russian Patients With Reproductive Issues (PRISMA)
AstraZeneca | Phase not applicable Completed Jun 17, 2024 | No interventions listed | Systemic Lupus Erythematosus |
Safety and Immunogenicity of a Booster Vaccination with an Adapted Vaccine
Hipra Scientific, S.L.U | Phase 2 / Phase 3 Completed Jun 17, 2024 | INTERVENTIONAL
| SARS-CoV2 Infection |
A Study to Evaluate Early Treatment Outcomes With Miebo™ in Subjects With Dry Eye Disease
Bausch & Lomb Incorporated | Phase 4 Completed Jun 17, 2024 | INTERVENTIONAL
| Dry Eye |
A Study to Investigate the Pharmacokinetics of Baxdrostat When Given Alone and in Combination With Itraconazole in Healthy Participants
AstraZeneca | Phase 1 Completed Jun 17, 2024 | INTERVENTIONAL
| Healthy Participants |
Evaluate the Efficacy and Safety of TTYP01 Tablets in the Treatment of Acute Ischemic Stroke
Shanghai Auzone Biological Technology Co., Ltd. | Phase 3 Completed Jun 17, 2024 | INTERVENTIONAL
| Acute Ischemic Stroke |
This is a Phase 1 Study in Which Healthy Volunteers and Participants with Chronic HBV Infection Will Receive HT-101 or Placebo and Will Be Assessed for Safety, Tolerability, Pharmacokinetics (PK), and Antiviral Activity
Suzhou HepaThera Biotech Co., Ltd. | Phase 1 Completed Jun 17, 2024 | INTERVENTIONAL
| Chronic Hepatitis B |
Efficacy of Oral Azacitidine Plus Best Supportive Care as Maintenance Therapy in Subjects With Acute Myeloid Leukemia (AML) in Complete Remission
Celgene | Phase 3 Completed Jun 18, 2024 | INTERVENTIONAL
| Leukemia, Myeloid, Acute |
GLOBAL ICON Stemless Shoulder System Post Market Clinical Follow Up Study
DePuy Orthopaedics | Phase not applicable Completed Jun 18, 2024 | INTERVENTIONAL
| Osteoarthritis
Post Traumatic Arthritis |
Feasibility Study of the Tendyne Mitral Valve System in Mitral Annular Calcification
Abbott Medical Devices | Phase not applicable Completed Jun 18, 2024 | INTERVENTIONAL
| Cardiovascular Diseases
Heart Valve Calcification
Heart Valve Diseases
Mitral Annular Calcification
Mitral Annulus Calcification
Mitral Insufficiency
Mitral Regurgitation
Mitral Valve Disease
Valve Heart Disease |
BilatEral Hypoglossal Nerve StimulaTion for TreatmEnt of ObstRuctive SLEEP Apnoea With and Without Complete Concentric Collapse
Nyxoah S.A. | Phase not applicable Completed Jun 18, 2024 | INTERVENTIONAL
| Obstructive Sleep Apnea |
Study of a High-Dose Aflibercept in Participants With Diabetic Eye Disease
Bayer | Phase 2 / Phase 3 Completed Jun 18, 2024 | INTERVENTIONAL
| Diabetic Macular Edema
Type 1 Diabetes Mellitus
Type 2 Diabetes Mellitus |
Study of a High-Dose Aflibercept in Participants With Diabetic Eye Disease
Regeneron Pharmaceuticals | Phase 2 / Phase 3 Completed Jun 18, 2024 | INTERVENTIONAL
| Diabetic Macular Edema
Type 1 Diabetes Mellitus
Type 2 Diabetes Mellitus |
A Study of Fruquintinib in Combination With Tislelizumab in Advanced Solid Tumors
BeiGene | Phase 1 / Phase 2 Completed Jun 18, 2024 | INTERVENTIONAL
| Colorectal Cancer
Endometrial Cancer
Solid Tumor, Unspecified, Adult
Triple Negative Breast Cancer |
A Study of Fruquintinib in Combination With Tislelizumab in Advanced Solid Tumors
Hutchmed | Phase 1 / Phase 2 Completed Jun 18, 2024 | INTERVENTIONAL
| Colorectal Cancer
Endometrial Cancer
Solid Tumor, Unspecified, Adult
Triple Negative Breast Cancer |
Medacta NextAR TKA Pivotal Trial
Medacta International SA | Phase not applicable Completed Jun 18, 2024 | INTERVENTIONAL
| Musculoskeletal Diseases |
A Study on How Vedolizumab is Given and the Outcomes for Adults With Inflammatory Bowel Disease (VARIETY-Belgium)
Takeda | Phase not applicable Completed Jun 18, 2024 | No interventions listed | Colitis, Ulcerative
Crohns Disease
Inflammatory Bowel Diseases |
Study to Evaluate the Safety and Tolerability of CC-92328 in Participants With Relapsed and/or Refractory Multiple Myeloma
Celgene | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Multiple Myeloma |
Clinical Evaluation of Fractional Bi-Polar Radio-Frequency for Symptoms of Vulvovaginal Atrophy
InMode MD Ltd. | Phase not applicable Completed Jun 18, 2024 | INTERVENTIONAL
| Vaginal Atrophy |
A Study in Male and Female Adult Patients With Atopic Dermatitis Treated With Dupilumab in Taiwan
Sanofi | Phase not applicable Completed Jun 18, 2024 | No interventions listed | Atopic Dermatitis |
Pivotal Trial for a Synthetic Tissue Substitute for Concealment of Artificial Ocular Implants
CorNeat Vision Ltd. | Phase not applicable Completed Jun 18, 2024 | INTERVENTIONAL
| Glaucoma Eye
Tissue Breakdown |
Clinical Efficacy and Safety Evaluation of Teneligliptin in Type 2 Diabetes Who Have Inadequate GlycemIc Control With Empaglyflozin 25 mg and Metformin
Handok Inc. | Phase 3 Completed Jun 18, 2024 | INTERVENTIONAL
| Type 2 Diabetes |
Post-transplant Flotetuzumab for AML
MacroGenics | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Leukemia, Myeloid, Acute |
Oral Rehydration Solutions in Healthy Adult Athletes
Abbott Nutrition | Phase not applicable Completed Jun 18, 2024 | INTERVENTIONAL
| Hydration |
A Randomized Controlled Trial to Compare the Glycemic Control, With or Without the Connected Solution, of Adult Type 2 Diabetic Participants on Basal Insulin in Taiwan
Sanofi | Phase not applicable Completed Jun 18, 2024 | INTERVENTIONAL
| Type 2 Diabetes Mellitus |
First in Human Study Evaluating Single Ascending Oral Doses of YCT-529 in Healthy Males
YourChoice Therapeutics, Inc. | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Male Contraception |
A Study to Evaluate the Safety and Immunogenicity of a Single Dose of Adsorbed Tetanus Vaccine in a Population Aged 18 Years and Over
CanSino Biologics Inc. | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Tetanus |
A Study to Evaluate the Effect of Jaktinib on QT/QTc Interval in Healthy Participants
Suzhou Zelgen Biopharmaceuticals Co.,Ltd | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Healthy Participants |
A Study to Assess the Potential for Gastric-pH Dependent Drug-Drug Interactions of BIIB122 With a Proton Pump Inhibitor in Healthy Participants
Biogen | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Healthy Volunteer |
A Study to Assess LY4100511 (DC-853) in Healthy Adult Participants
DICE Therapeutics, Inc., a wholly owned subsidiary of Eli Lilly and Company | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Healthy |
A Study to Assess LY4100511 (DC-853) in Healthy Adult Participants
Eli Lilly and Company | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Healthy |
Effect of Topical PlexoZome® Levagen® Spray on Relief of Post Exercise Knee Joint Pain
RDC Clinical Pty Ltd | Phase 4 Completed Jun 18, 2024 | INTERVENTIONAL
| Knee Joint Pain |
Effect of Erythromycin on the Absorption, Metabolism and Elimination of CHF6001 in Healthy Volunteers
Chiesi Farmaceutici S.p.A. | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Chronic Obstructive Pulmonary Disease |
A Study to Assess the Safety and Efficacy of a Sunscreen on Acne-Prone Skin With Inflammatory and Non-Inflammatory Acne Vulgaris
JNTL Consumer Health (Brasil) LTDA | Phase not applicable Completed Jun 18, 2024 | INTERVENTIONAL
| Acne Vulgaris |
Safety, Tolerability and Pharmacokinetics of LNK01004 Ointment in Adults With Atopic Dermatitis.
Lynk Pharmaceuticals Co., Ltd | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Atopic Dermatitis |
Phase 1 Study of HT-102 Administered Subcustaneously in Healthy Participants and Patients with Chronic Hepatitis B for Safety, Tolerability, Pharmacokinetics (PK), and Antiviral Activity (only in Participants with Chronic HBV Infection)
Suzhou HepaThera Biotech Co., Ltd. | Phase 1 Completed Jun 18, 2024 | INTERVENTIONAL
| Chronic Hepatitis B |
A Real-world Study of the Effectiveness of Tisagenlecleucel in Acute Lymphoblastic Leukemia Patients
Novartis Pharmaceuticals | Phase not applicable Completed Jun 18, 2024 | No interventions listed | Acute Lymphoblastic Leukemia |
The PROMISE Study: Duavee in Women With DCIS
Pfizer | Phase 2 Completed Jun 19, 2024 | INTERVENTIONAL
| Ductal Breast Carcinoma In Situ
Postmenopausal |
Study of Bone Growth in the Sacroiliac Joint After Minimally Invasive Surgery With Titanium Implants
SI-BONE, Inc. | Phase not applicable Completed Jun 19, 2024 | INTERVENTIONAL
| Sacroiliac Dysfunction |
China S3: Safety and Effectiveness of Edwards Lifesciences SAPIEN 3 THV in the Chinese Population
Edwards Lifesciences | Phase not applicable Completed Jun 19, 2024 | INTERVENTIONAL
| Aortic Regurgitation
Aortic Stenosis
Symptomatic Aortic Stenosis |
Clinical Study Comparing Two Models of a Travoprost Intraocular Implant
Glaukos Corporation | Phase 3 Completed Jun 19, 2024 | INTERVENTIONAL
| Glaucoma, Open-Angle
Ocular Hypertension |
Clinical Study Comparing Two Models of a Travoprost Intraocular Implant
Glaukos Corporation | Phase 3 Completed Jun 19, 2024 | INTERVENTIONAL
| Glaucoma, Open-Angle
Ocular Hypertension |
An Open, Study to Assess the Safety of RGTA® (OTR4132) in Patients With Acute Ischemic Stroke (AIS)
Euraxi Pharma | Phase not applicable Completed Jun 19, 2024 | INTERVENTIONAL
| Stroke, Acute |
An Open, Study to Assess the Safety of RGTA® (OTR4132) in Patients With Acute Ischemic Stroke (AIS)
Organ, Tissue, Regeneration, Repair and Replacement | Phase not applicable Completed Jun 19, 2024 | INTERVENTIONAL
| Stroke, Acute |
Etrasimod Versus Placebo for the Treatment of Moderately Active Ulcerative Colitis
Arena is a wholly owned subsidiary of Pfizer | Phase 2 Completed Jun 19, 2024 | INTERVENTIONAL
| Ulcerative Colitis |
Etrasimod Versus Placebo for the Treatment of Moderately Active Ulcerative Colitis
Pfizer | Phase 2 Completed Jun 19, 2024 | INTERVENTIONAL
| Ulcerative Colitis |
Safety and Pharmacokinetics of Fluzoparib in Healthy Subjects and Those With Impaired Liver Function
Jiangsu HengRui Medicine Co., Ltd. | Phase 1 Completed Jun 19, 2024 | INTERVENTIONAL
| Healthy Subjects
Hepatic Impairment |
Efficacy and Safety Study of a New Therapeutic Strategy in the Treatment of Extended-Disease Small-Cell Lung Cancer
YGHEA, CRO Division of Ecol Studio spa | Phase 2 Completed Jun 19, 2024 | INTERVENTIONAL
| Small Cell Lung Cancer |
A Study of Trastuzumab Emtansine in Combination With Atezolizumab or Placebo as a Treatment for Participants With Human Epidermal Growth Factor 2 (HER2)-Positive and Programmed Death-ligand 1 (PD-L1)-Positive Locally Advanced (LABC) or Metastatic Breast Cancer (MBC)
Hoffmann-La Roche | Phase 3 Completed Jun 19, 2024 | INTERVENTIONAL
| Metastatic Breast Cancer |
PF-07284892 in Participants With Advanced Solid Tumors
Pfizer | Phase 1 Completed Jun 19, 2024 | INTERVENTIONAL
| Solid Tumor |
Efficacy and Safety Study of Orvepitant for Chronic Cough in Patients With Idiopathic Pulmonary Fibrosis
Nerre Therapeutics Ltd. | Phase 2 Completed Jun 19, 2024 | INTERVENTIONAL
| Cough
Idiopathic Pulmonary Fibrosis |
Efficacy and Safety Study of Orvepitant for Chronic Cough in Patients With Idiopathic Pulmonary Fibrosis
Pharm-Olam International | Phase 2 Completed Jun 19, 2024 | INTERVENTIONAL
| Cough
Idiopathic Pulmonary Fibrosis |
Efficacy, Safety and Pharmacokinetics Study of CPL500036 (PDE10A Inhibitor) in Patients With Schizophrenia
Celon Pharma SA | Phase 2 Completed Jun 19, 2024 | INTERVENTIONAL
| Schizophrenia |
Phase 3 Safety and Efficacy Study of CTAP101 Extended-release Capsules in Children With Secondary Hyperparathyroidism
OPKO Health, Inc. | Phase 3 Completed Jun 19, 2024 | INTERVENTIONAL
| Chronic Kidney Disease stage3
Chronic Kidney Disease stage4
Secondary Hyperparathyroidism
Vitamin d Deficiency |
A Study to Evaluate the Effects of Cenicriviroc Mesylate on Arterial Inflammation in People Living With HIV
AbbVie | Phase 2 Completed Jun 19, 2024 | INTERVENTIONAL
| Elevated Cardiovascular Risk
HIV-1-infection |
A Study to Investigate Different Formulations of S-337395 in Healthy Adult Participants
Shionogi | Phase 1 Completed Jun 19, 2024 | INTERVENTIONAL
| Healthy Volunteers |
A Study to Evaluate the Relative Bioavailability and Food Effect of a New Tablet Formulation and Strength of Suzetrigine
Vertex Pharmaceuticals Incorporated | Phase 1 Completed Jun 19, 2024 | INTERVENTIONAL
| Pain |
Clinical Trial to Evaluate the Efficacy and Safety of DW1026C1 or DW1026C2
Daewon Pharmaceutical Co., Ltd. | Phase 3 Completed Jun 19, 2024 | INTERVENTIONAL
| Type 2 Diabetes |
High Dose Cytarabine Followed by Pembrolizumab in Relapsed/Refractory AML
Merck Sharp & Dohme LLC | Phase 2 Completed Jun 20, 2024 | INTERVENTIONAL
| Acute Myeloid Leukemia, in Relapse |
Trial to Assess Safety and Efficacy of Lenvatinib (18 mg vs. 14 mg) in Combination With Everolimus in Participants With Renal Cell Carcinoma
Eisai Inc. | Phase 2 Completed Jun 20, 2024 | INTERVENTIONAL
| Renal Cell Carcinoma |
Trial to Assess Safety and Efficacy of Lenvatinib (18 mg vs. 14 mg) in Combination With Everolimus in Participants With Renal Cell Carcinoma
Merck Sharp & Dohme LLC | Phase 2 Completed Jun 20, 2024 | INTERVENTIONAL
| Renal Cell Carcinoma |
Lumbar Belt Benefit Compared to the Usual Care in the Treatment of Non-specific Low Back Pain
RCTs | Phase not applicable Completed Jun 20, 2024 | INTERVENTIONAL
| Low Back Pain |
Lumbar Belt Benefit Compared to the Usual Care in the Treatment of Non-specific Low Back Pain
Thuasne | Phase not applicable Completed Jun 20, 2024 | INTERVENTIONAL
| Low Back Pain |
Mitoxantrone Hydrochloride Liposome Injection, Bortezomib and Dexamethasone in the Treatment of R/R MM
CSPC Zhongnuo Pharmaceutical (Shijiazhuang) Co., Ltd. | Recruiting
Phase 1 Past estimated: Jun 20, 2024 | INTERVENTIONAL
| Relapsed or Refractory Multiple Myeloma |
A Study of Chinese Adults With Lymphoma
Takeda | Phase not applicable Completed Jun 20, 2024 | No interventions listed | Lymphoma |
Evaluation of MoodRing on Improving the Quality of Depression Management in Adolescents
NuRelm, Inc. | Phase not applicable Completed Jun 20, 2024 | INTERVENTIONAL
| Depression in Adolescence |
Efficacy and Safety of Inclisiran as Monotherapy in Patients With Primary Hypercholesterolemia Not Receiving Lipid-lowering Therapy.
Novartis Pharmaceuticals | Phase 3 Completed Jun 20, 2024 | INTERVENTIONAL
| Hypercholesterolemia |
[14 C] Study on Mass Balance of TY-9591 in Healthy Chinese Subjects
TYK Medicines, Inc | Phase 1 Completed Jun 20, 2024 | INTERVENTIONAL
| Healthy Volunteers |
SWC for Treatment of Superficial Partial-Thickness Burns
Synedgen, Inc. | Phase not applicable Completed Jun 20, 2024 | INTERVENTIONAL
| Burn Wounds |
Feasibility Study for Instant Cardiac Diagnostic and Post-operative Risk Assessment with the LynxPatch Device
Lynx Health Science GmbH | Phase not applicable Completed Jun 20, 2024 | OBSERVATIONAL
| Cardiovascular Diseases
Heart Diseases |
Feasibility Study for Instant Cardiac Diagnostic and Post-operative Risk Assessment with the LynxPatch Device
Meditrial Europe Ltd. | Phase not applicable Completed Jun 20, 2024 | OBSERVATIONAL
| Cardiovascular Diseases
Heart Diseases |
A Study of EDG-7500 in Healthy Adults
Edgewise Therapeutics, Inc. | Phase 1 Completed Jun 20, 2024 | INTERVENTIONAL
| Healthy Subjects |
EX-PRESS® P-50 and EX-PRESS® P-200
Alcon Research | Phase not applicable Completed Jun 20, 2024 | OBSERVATIONAL
| Glaucoma |
AIMIGo 12L ECG Synthesis Software Pivotal Study for Arrythmia Detection
HeartBeam, Inc. | Phase not applicable Completed Jun 20, 2024 | INTERVENTIONAL
| Arrhythmias, Cardiac
Atrial Fibrillation
Atrial Flutter
Bradycardia
Tachycardia |
Treatment of Adults With Acute Bronchitis in the Primary Healthcare Setting
Neopharm Bulgaria Ltd. | Phase not applicable Completed Jun 20, 2024 | INTERVENTIONAL
| Acute Bronchitis |
Clinical Study to Evaluate the Anti-sensitivity Efficacy of a Stannous Fluoride Toothpaste in a Population of Dentin Hypersensitivity Sufferers
HALEON | Phase not applicable Completed Jun 20, 2024 | INTERVENTIONAL
| Dentin Sensitivity |
A Trial to Evaluate the Effect of Esomeprazole on the Pharmacokinetics of Emraclidine in Healthy Adult Participants
Cerevel Therapeutics, LLC | Phase 1 Completed Jun 20, 2024 | INTERVENTIONAL
| Healthy Participants |
A Study to Evaluate the Safety and Efficacy of DSFNC1 Soft Hydrophilic Contact Lenses and SiHy Color Soft Hydrophilic Contact Lenses
BenQ Materials Corporation | Phase not applicable Completed Jun 20, 2024 | INTERVENTIONAL
| Myopia |
A Clinical Trial to Investigate the Safety and Efficacy of D-Mannose on Symptoms of Urinary Tract Infection in Women
KEB Nutraceuticals USA, Inc. | Phase 2 Completed Jun 20, 2024 | INTERVENTIONAL
| Urinary Tract Infection(UTI) |
A Clinical Trial to Investigate the Safety and Efficacy of D-Mannose on Symptoms of Urinary Tract Infection in Women
KGK Science Inc. | Phase 2 Completed Jun 20, 2024 | INTERVENTIONAL
| Urinary Tract Infection(UTI) |
Study of SAL-0951 Compared to Recombinant Human Erythropoietin in Anemic Patients Receiving Maintenance Hemodialysis
Shenzhen Salubris Pharmaceuticals Co., Ltd. | Phase 3 Completed Jun 20, 2024 | INTERVENTIONAL
| Anemia |
A Study of the Effectiveness and Safety of Nivolumab Compared to Bevacizumab and of Nivolumab With or Without Ipilimumab in Glioblastoma Patients
Bristol-Myers Squibb | Phase 3 Completed Jun 21, 2024 | INTERVENTIONAL
| Recurrent Glioblastoma |
A Trial to Evaluate the Efficacy and Safety of Tafasitamab With Bendamustine (BEN) Versus Rituximab (RTX) With BEN in Adult Patients With Relapsed or Refractory Diffuse Large B-cell Lymphoma (DLBCL)
Incyte Corporation | Phase 2 / Phase 3 Completed Jun 21, 2024 | INTERVENTIONAL
| Diffuse Large B-cell Lymphoma |
Neoadjuvant Durvalumab and Tremelimumab Plus Radiation for High Risk Soft-Tissue Sarcoma
AstraZeneca | Phase 1 / Phase 2 Completed Jun 21, 2024 | INTERVENTIONAL
| Soft Tissue Sarcoma |
A Phase 1/2 Study of FS118 in Patients With Advanced Malignancies
invoX Pharma Limited | Phase 1 / Phase 2 Completed Jun 21, 2024 | INTERVENTIONAL
| Advanced Cancer
Metastatic Cancer
Squamous Cell Carcinoma of Head and Neck |
Mepolizumab for COPD Hospital Eosinophilic Admissions Pragmatic Trial
GlaxoSmithKline | Phase 2 / Phase 3 Completed Jun 21, 2024 | INTERVENTIONAL
| COPD
Eosinophilia |
Efficacy and Safety of IBI310 Combined with Sintilimab in Patients with Advanced Hepatocellular Carcinoma
Innovent Biologics (Suzhou) Co. Ltd. | Phase 1 Completed Jun 21, 2024 | INTERVENTIONAL
| Advanced Hepatocellular Carcinoma |
LUMINOS-101: Lerapolturev (PVSRIPO) and Pembrolizumab in Patients With Recurrent Glioblastoma
Istari Oncology, Inc. | Phase 2 Completed Jun 21, 2024 | INTERVENTIONAL
| Brain Tumor
Glioblastoma
Recurrent Glioblastoma
Supratentorial Glioblastoma |
Efficacy and Safety of MK-8189 in Participants With an Acute Episode of Schizophrenia (MK-8189-008)
Merck Sharp & Dohme LLC | Phase 2 Completed Jun 21, 2024 | INTERVENTIONAL
| Schizophrenia |
Autonomous Robotics for Transcatheter Cardiac Procedures
Artiness SRL | Phase not applicable Completed Jun 21, 2024 | OBSERVATIONAL
| Heart Valve Diseases
Mitral Valve Disease
Structural Heart Abnormality |
Specified Drug-use Survey on Abilify Prolonged Release Aqueous Suspension for IM Injection (Prevention of Relapse of Mood Episodes in BP)
Otsuka Pharmaceutical Co., Ltd. | Phase not applicable Completed Jun 21, 2024 | OBSERVATIONAL
| Bipolar Disorder I |
Study of ASP7517 Alone and With Pembrolizumab in Participants With Advanced Solid Tumors Expressing WT1 Antigen
Astellas Pharma Global Development, Inc. | Phase 1 / Phase 2 Completed Jun 21, 2024 | INTERVENTIONAL
| Advanced Cancer
Advanced Malignancies |
Radiotherapy in Combination With Atezolizumab Prior to Surgical Resection for HPV Unrelated HNSCC
Genentech, Inc. | Phase 1 Completed Jun 21, 2024 | INTERVENTIONAL
| Squamous Cell Carcinoma of Head and Neck |
A 12-Week Crossover Study to Assess the Efficacy, Safety and Tolerability of L1-79 in Subjects Aged 12-21 Years With Autism Spectrum Disorder
Yamo Pharmaceuticals LLC | Phase 2 Completed Jun 21, 2024 | INTERVENTIONAL
| Autism
Autism Spectrum Disorder |
Evaluation of the MiniMed™ 780G System in Type 1 Adult and Pediatric Subjects Utilizing Insulin Fiasp®
Medtronic Diabetes | Phase not applicable Completed Jun 21, 2024 | INTERVENTIONAL
| Type 1 Diabetes |
Validation Study of a Patch-based PSG System
Onera BV | Phase not applicable Past estimated: Jun 21, 2024 | INTERVENTIONAL
| Sleep Disorder |
Doxil/Caelyx BE Study
Baxter Healthcare Corporation | Phase 1 Completed Jun 21, 2024 | INTERVENTIONAL
| AIDS-related Kaposi Sarcoma
Metastatic Breast Cancer
Multiple Myeloma
Ovarian Cancer |
Study of StimRouter for Chronic Knee OA Pain
Bioventus LLC | Phase not applicable Completed Jun 21, 2024 | OBSERVATIONAL
| Osteoarthritis, Knee |
Closed Loop Glucose Control in Patients With Type 1 and Type 2 Diabetes
Ideal Medical Technologies | Phase not applicable Completed Jun 21, 2024 | INTERVENTIONAL
| Diabetes Mellitus, Type 1
Diabetes Mellitus, Type 2 |
Effects of Pressure-controlled Intermittent Coronary Sinus Occlusion (PiCSO) Therapy on miRNAs Expression and Absolute Microvascular Resistance by Continuous Thermodilution in ST-segment Elevation Myocardial Infarction (STEMI) Patients: PiCSO-STEMI TRIAL
Miracor Medical SA | Phase not applicable Completed Jun 21, 2024 | INTERVENTIONAL
| Anterior Myocardial Infarction |
Phase 2 Study to Evaluate RPT193 in Adults With Moderate to Severe T2-high Asthma
RAPT Therapeutics, Inc. | Phase 2 Completed Jun 21, 2024 | INTERVENTIONAL
| Asthma |
A Phase I Study of BR108 in Hematological Malignancies
BioRay Pharmaceutical Co., Ltd. | Phase 1 Completed Jun 21, 2024 | INTERVENTIONAL
| Hematologic Malignancy |
A Study to Evaluate the Safety, Pharmacokinetics and Efficacy of AK120 in Subjects With Atopic Dermatitis
Akeso | Phase 1 / Phase 2 Completed Jun 21, 2024 | INTERVENTIONAL
| Atopic Dermatitis |
Pilot Randomized Study of RD-X19 Tx Device in Subjects With PCC (Long Covid) in the Outpatient Setting
EmitBio Inc. | Phase not applicable Completed Jun 21, 2024 | INTERVENTIONAL
| Post COVID-19 Condition (PCC) |
A Study to Evaluate the Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of K-757 and K-833 in Overweight/Obese Patients With Type 2 Diabetes
Kallyope Inc. | Phase 1 Completed Jun 21, 2024 | INTERVENTIONAL
| Obesity
Type 2 Diabetes Mellitus (T2DM) |
Studying Glucose and Appetite Response With Alternatives to Soda Pop
Olipop, PBC | Phase not applicable Completed Jun 21, 2024 | INTERVENTIONAL
| Healthy Nutrition |
To Compare the Safety and PK/ PD Characteristics of ADC189 Between Hepatic Impairment and Normal Hepatic Function Subjects
Jiaxing AnDiCon Biotech Co.,Ltd | Phase 1 Completed Jun 21, 2024 | INTERVENTIONAL
| Influenza Type A
Influenza Type B |
A Study to Evaluate the Drug-Drug Interaction Between Enlicitide Decanoate (MK-0616) and Alendronate in Healthy Adult Participants (MK-0616-027)
Merck Sharp & Dohme LLC | Phase 1 Completed Jun 21, 2024 | INTERVENTIONAL
| Healthy |
Evaluating the Effectiveness of a Digital Therapeutic (Reviga) for People With Stress or Burnout
Gaia AG | Phase not applicable Completed Jun 22, 2024 | INTERVENTIONAL
| Burnout
Stress |
A Clinical Study to Evaluate the Anti-Sensitivity Efficacy of a Stannous Fluoride Toothpaste in a Population of Dentin Hypersensitivity Sufferers
HALEON | Phase not applicable Completed Jun 23, 2024 | INTERVENTIONAL
| Dentin Sensitivity |
Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of (PEG)-BHD1028
EncuraGen, Inc | Phase 1 Completed Jun 23, 2024 | INTERVENTIONAL
| Insulin Resistance |
A Study of MK-8507 and Islatravir (MK-8591) in Healthy Adult Participants (MK-8507-016)
Merck Sharp & Dohme LLC | Phase 1 Completed Jun 23, 2024 | INTERVENTIONAL
| Healthy |
A Study of the Bruton's Tyrosine Kinase Inhibitor Ibrutinib Given in Combination With Bendamustine and Rituximab in Patients With Newly Diagnosed Mantle Cell Lymphoma
Janssen Research & Development, LLC | Phase 3 Completed Jun 24, 2024 | INTERVENTIONAL
| Mantle Cell Lymphoma |
A Study of the Bruton's Tyrosine Kinase Inhibitor Ibrutinib Given in Combination With Bendamustine and Rituximab in Patients With Newly Diagnosed Mantle Cell Lymphoma
Pharmacyclics LLC. | Phase 3 Completed Jun 24, 2024 | INTERVENTIONAL
| Mantle Cell Lymphoma |
Atezolizumab in Combination With Entinostat and Bevacizumab in Patients With Advanced Renal Cell Carcinoma
Genentech, Inc. | Phase 1 / Phase 2 Completed Jun 24, 2024 | INTERVENTIONAL
| Metastatic Cancer
Renal Cancer |
Atezolizumab in Combination With Entinostat and Bevacizumab in Patients With Advanced Renal Cell Carcinoma
Syndax Pharmaceuticals | Phase 1 / Phase 2 Completed Jun 24, 2024 | INTERVENTIONAL
| Metastatic Cancer
Renal Cancer |
Oral STAT3 Inhibitor, TTI-101, in Patients with Advanced Cancers
Tvardi Therapeutics, Incorporated | Phase 1 Completed Jun 24, 2024 | INTERVENTIONAL
| Advanced Cancer
Breast Cancer
Colorectal Cancer
Gastric Adenocarcinoma
Head and Neck Squamous Cell Carcinoma
Hepatocellular Cancer
Melanoma
Non Small Cell Lung Cancer |
Study to Assess AFM24 in Advanced Solid Cancers
Affimed GmbH | Phase 1 / Phase 2 Completed Jun 24, 2024 | INTERVENTIONAL
| Advanced Solid Tumor |
Hemiverse Shoulder Prosthesis
41Hemiverse AG | Phase not applicable Completed Jun 24, 2024 | INTERVENTIONAL
| Hemiarthroplasty
Rotator Cuff Tears |
A Study of AMG 340 in Subjects With Metastatic Castrate-Resistant Prostate Carcinoma
Amgen | Phase 1 Completed Jun 24, 2024 | INTERVENTIONAL
| Metastatic Castration-resistant Prostate Cancer |
A Safety and Efficacy Study of Anti-inflammatory (Canakinumab) and Cartilage Stimulating (LNA043) Drugs Injected Into the Knee Joint of Participants With Knee Osteoarthritis (OA)
Novartis Pharmaceuticals | Phase 2 Completed Jun 24, 2024 | INTERVENTIONAL
| Knee Osteoarthritis |
PRV-3279-2a Trial in Systemic Lupus
Provention Bio, a Sanofi Company | Phase 2 Completed Jun 24, 2024 | INTERVENTIONAL
| Systemic Lupus Erythematosus |
Effects of Edoxaban on Platelet Aggregation
Daiichi Sankyo | Phase 2 / Phase 3 Completed Jun 24, 2024 | INTERVENTIONAL
| AMI
SCAD |
Safety, Effectiveness and Quality of Life of Trastuzumab Deruxtecan in Patients with HER2+ Breast Cancer
Daiichi Sankyo France | Phase not applicable Completed Jun 24, 2024 | No interventions listed | HER2-positive Breast Cancer |
Human Milk Oligosaccharides (HMOs) for Irritable Bowel Syndrome (IBS)
DSM Nutritional Products, Inc. | Phase not applicable Completed Jun 24, 2024 | INTERVENTIONAL
| IBS - Irritable Bowel Syndrome
Irritable Bowel Syndrome |
A Study of Hepalatide Combined With TAF and PEG-IFN as Finite Treatment of Chronic Hepatitis B Patients
Shanghai HEP Pharmaceutical Co., Ltd. | Phase 2 Completed Jun 24, 2024 | INTERVENTIONAL
| Chronic Hepatitis B |
A Study Testing the Superiority of CHF 1535 pMDI 800/24µg Total Daily Dose Compared With CHF 718 pMDI 800µg Total Daily Dose in Adults With Asthma on Medium or High-Dose Inhaled Corticosteroid
Chiesi Farmaceutici S.p.A. | Phase 3 Completed Jun 24, 2024 | INTERVENTIONAL
| Asthma |
A Study to Learn About Sickle Cell Disease In Adult Patients
Pfizer | Phase not applicable Completed Jun 24, 2024 | OBSERVATIONAL
| Sickle Cell Disease |
Real-world Dapagliflozin Treatment in Patients With Heart Failure in Portugal (EVOLUTION-HF)
AstraZeneca | Phase not applicable Completed Jun 24, 2024 | No interventions listed | Heart Failure |
QA108 Phase II Study in Subjects With Intermediate Age-Related Macular Degeneration
Smilebiotek Zhuhai Limited | Phase 2 Completed Jun 24, 2024 | INTERVENTIONAL
| Intermediate Age-Related Macular Degeneration |
Satralizumab in Aneurysmal Subarachnoid Hemorrhage
Genentech, Inc. | Phase 1 Completed Jun 24, 2024 | INTERVENTIONAL
| Aneurysmal Subarachnoid Hemorrhage
Delayed Cerebral Ischemia |
Remote Monitoring of Vital Signs With Telehealth to Prevent Readmissions for Dysglycemia
DexCom, Inc. | Phase not applicable Completed Jun 24, 2024 | INTERVENTIONAL
| Dysglycemia |
Pimavanserin for Sleep in Parkinson Disease
ACADIA Pharmaceuticals Inc. | Phase 1 Completed Jun 24, 2024 | INTERVENTIONAL
| Parkinson Disease |
Study of mRNA-1010 Seasonal Influenza Vaccine in Adults
ModernaTX, Inc. | Phase 3 Completed Jun 24, 2024 | INTERVENTIONAL
| Seasonal Influenza |
Evaluation of an Herbal Extract on Sleep Parameters
Aventure AB | Phase not applicable Completed Jun 24, 2024 | INTERVENTIONAL
| Sleep Onset Latency |
A Study to Characterize the Prevalence of Overweight and Obesity Among Patients With Established Cardiovascular Disease
Novo Nordisk A/S | Phase not applicable Completed Jun 24, 2024 | OBSERVATIONAL
| Obesity
Overweight |
INHALE-3: Afrezza® Combined With Insulin Degludec Versus Usual Care in Adults With Type 1 Diabetes
Mannkind Corporation | Phase 4 Completed Jun 24, 2024 | INTERVENTIONAL
| Diabetes Mellitus, Type 1 |
Proof of Concept Treatment Study of Orally Administered VH4004280 or VH4011499 in HIV-1 Infected Adults
ViiV Healthcare | Phase 2 Completed Jun 24, 2024 | INTERVENTIONAL
| HIV Infections |
Wearable Auscultation Device Validation in Children
Strados Labs, Inc. | Phase not applicable Completed Jun 24, 2024 | OBSERVATIONAL
| Asthma
Asthma in Children
Pediatric Asthma
Wheezing |
A Positron-emission Tomography Study to Determine Brain Exposure of [11C]Savolitinib in Healthy Volunteers
AstraZeneca | Phase 1 Completed Jun 24, 2024 | INTERVENTIONAL
| Healthy Volunteers |
An Observational Study to Learn More About Vericiguat Treatment Patterns and Its Safety in People With Chronic Heart Failure With Reduced Ejection Fraction in Routine Medical Care in the United States
Bayer | Phase not applicable Completed Jun 24, 2024 | OBSERVATIONAL
| Chronic Heart Failure With Reduced Ejection Fraction |
Evaluation of the Clinical Performance of Daily Disposable Silicone Hydrogel Multifocal Toric Contact Lenses
Johnson & Johnson Vision Care, Inc. | Phase not applicable Completed Jun 24, 2024 | INTERVENTIONAL
| Visual Acuity |
A Randomized Triple-Blind Placebo-Controlled Study to Evaluate the Effects of a Supplement on Lower Urinary Tract Symptoms in Men.
Citruslabs | Phase not applicable Completed Jun 24, 2024 | INTERVENTIONAL
| Benign Prostatic Hyperplasia
Erectile Dysfunction
Lower Urinary Tract Symptoms |
A Randomized Triple-Blind Placebo-Controlled Study to Evaluate the Effects of a Supplement on Lower Urinary Tract Symptoms in Men.
Optimale | Phase not applicable Completed Jun 24, 2024 | INTERVENTIONAL
| Benign Prostatic Hyperplasia
Erectile Dysfunction
Lower Urinary Tract Symptoms |
A Single Group Study to Evaluate the Effects of a Probiotic Supplement on Vaginal Health.
Citruslabs | Phase not applicable Completed Jun 24, 2024 | INTERVENTIONAL
| Vaginal Health
Women's Health |
A Single Group Study to Evaluate the Effects of a Probiotic Supplement on Vaginal Health.
Love Wellness | Phase not applicable Completed Jun 24, 2024 | INTERVENTIONAL
| Vaginal Health
Women's Health |
Randomized Placebo Controlled Clinical Study of an Oral Product in Subjects With Menopausal Symptoms
Olly, PBC | Phase not applicable Completed Jun 24, 2024 | INTERVENTIONAL
| Menopausal Symptoms |
Pembrolizumab and Trametinib in Treating Patients With Stage IV Non-Small Cell Lung Cancer and KRAS Gene Mutations
Merck Sharp & Dohme LLC | Phase 1 Completed Jun 25, 2024 | INTERVENTIONAL
| KRAS Gene Mutation
Metastatic Non-Squamous Non-Small Cell Lung Carcinoma
Recurrent Non-Squamous Non-Small Cell Lung Carcinoma
Stage IV Non-Small Cell Lung Cancer AJCC v7 |
Pembrolizumab and Trametinib in Treating Patients With Stage IV Non-Small Cell Lung Cancer and KRAS Gene Mutations
Novartis | Phase 1 Completed Jun 25, 2024 | INTERVENTIONAL
| KRAS Gene Mutation
Metastatic Non-Squamous Non-Small Cell Lung Carcinoma
Recurrent Non-Squamous Non-Small Cell Lung Carcinoma
Stage IV Non-Small Cell Lung Cancer AJCC v7 |
M7824 Versus Pembrolizumab as a First-line (1L) Treatment in Participants With Programmed Death-ligand 1 (PD-L1) Expressing Advanced Non-small Cell Lung Cancer (NSCLC)
EMD Serono Research & Development Institute, Inc. | Phase 3 Completed Jun 25, 2024 | INTERVENTIONAL
| Non-small Cell Lung Cancer |
M7824 Versus Pembrolizumab as a First-line (1L) Treatment in Participants With Programmed Death-ligand 1 (PD-L1) Expressing Advanced Non-small Cell Lung Cancer (NSCLC)
Merck KGaA, Darmstadt, Germany | Phase 3 Completed Jun 25, 2024 | INTERVENTIONAL
| Non-small Cell Lung Cancer |
Clinical Outcomes Associated With the Use of ViviGen® for the Treatment of Lumbar Degenerative Disc Disease
DePuy Synthes | Phase not applicable Completed Jun 25, 2024 | INTERVENTIONAL
| Degenerative Disc Disease
Degenerative Spondylolisthesis
Lumbar Disc Disease
Lumbar Radiculopathy
Lumbar Spinal Stenosis
Lumbar Spondylolisthesis |
FOCUS: A Phase I/II First in Human Study to Evaluate the Safety and Efficacy of GT005 Administered in Subjects With Dry AMD
Gyroscope Therapeutics Limited | Phase 1 / Phase 2 Completed Jun 25, 2024 | INTERVENTIONAL
| Dry Age-related Macular Degeneration
Eye Diseases
Geographic Atrophy
Macular Atrophy
Macular Degeneration
Retinal Degeneration
Retinal Disease |
FOCUS: A Phase I/II First in Human Study to Evaluate the Safety and Efficacy of GT005 Administered in Subjects With Dry AMD
Novartis Pharmaceuticals | Phase 1 / Phase 2 Completed Jun 25, 2024 | INTERVENTIONAL
| Dry Age-related Macular Degeneration
Eye Diseases
Geographic Atrophy
Macular Atrophy
Macular Degeneration
Retinal Degeneration
Retinal Disease |
A Study of Nivolumab or Placebo in Combination With Docetaxel in Men With Advanced Castration-resistant Prostate Cancer
Bristol-Myers Squibb | Phase 3 Completed Jun 25, 2024 | INTERVENTIONAL
| Prostate Cancer |
Investigation of Chronic Obstructive Pulmonary Disease (COPD) Phenotypes and Endotypes in China
GlaxoSmithKline | Phase not applicable Completed Jun 25, 2024 | OBSERVATIONAL
| Pulmonary Disease, Chronic Obstructive |
A Phase I Clinical Trial of Y150 in the Treatment of Relapsed or Refractory Multiple Myeloma
Wuhan YZY Biopharma Co., Ltd. | Phase 1 Completed Jun 25, 2024 | INTERVENTIONAL
| Relapsed or Refractory Multiple Myeloma |
BAT7104 in Patients With Advanced Solid Tumours
Bio-Thera Solutions | Phase 1 Completed Jun 25, 2024 | INTERVENTIONAL
| Patients With Advanced Solid Tumors |
BAT7104 in Patients With Advanced Solid Tumours
Emerald Clinical Inc. | Phase 1 Completed Jun 25, 2024 | INTERVENTIONAL
| Patients With Advanced Solid Tumors |
A Study of CM310 in Patients With Chronic Rhinosinusitis With Nasal Polyposis (CROWNS-2)
Keymed Biosciences Co.Ltd | Phase 3 Completed Jun 25, 2024 | INTERVENTIONAL
| Chronic Rhinosinusitis With Nasal Polyposis |
BTL-785F Device for Non-invasive Reduction of Wrinkles and Overall Facial Improvement
BTL Industries Ltd. | Phase not applicable Completed Jun 25, 2024 | INTERVENTIONAL
| Wrinkle |
Clinical Research Study to Learn About the Effect and Safety of Different Doses of FE 999302 When Given as a Single Dose for Final Development of the Eggs After Ovarian Stimulation
Ferring Pharmaceuticals | Phase 2 Past estimated: Jun 25, 2024 | INTERVENTIONAL
| Infertility |
Treatment of Ano-perineal Fistulas Resistant to Surgical Treatment with a Gel and Plug of Wharton's Jelly
TBF Genie Tissulaire | Phase 1 Completed Jun 25, 2024 | INTERVENTIONAL
| Anal Fistula |
SHR-2004 for the Prevention of Venous Thromboembolism in Patients Undergoing Total Knee Arthroplasty
Beijing Suncadia Pharmaceuticals Co., Ltd | Phase 2 Completed Jun 25, 2024 | INTERVENTIONAL
| Prevention of Venous Thromboembolism After Knee Arthroplasty |
Efficacy of OmegaBoost to Increase Omega-3 Levels in Healthy Individuals
Guardion Health Sciences, Inc. | Phase not applicable Past estimated: Jun 25, 2024 | INTERVENTIONAL
| Age-Related Macular Degeneration
Dry Eye Syndromes
Omega 3 Index |
A Post-market Study to Evaluate the Effects of Sodium Hyaluronate Based Eye Drops in Patients Affected by Dry Eye Disease
Montefarmaco OTC SpA | Phase not applicable Completed Jun 25, 2024 | INTERVENTIONAL
| Opthalomology/Dry Eye |
Transthyretin Amyloidosis Cardiomyopathy in Patients With HFpEF in Russia
AstraZeneca | Phase not applicable Completed Jun 25, 2024 | No interventions listed | Transthyretin Amyloidosis Cardiomyopathy, Heart Failure With Preserved Ejection Fraction |
Efficacy and Safety of Probiotic Products for Digestive Health
Wecare Probiotics Co., Ltd. | Phase not applicable Past estimated: Jun 25, 2024 | INTERVENTIONAL
| Healthy Adult |
Clinical Experimental Study to Investigate Specific Features of ClampArt®
Profil Institut für Stoffwechselforschung GmbH | Phase not applicable Completed Jun 26, 2024 | INTERVENTIONAL
| Diabetes Mellitus |
A Study of Avelumab With Axitinib Versus Sunitinib In Advanced Renal Cell Cancer (JAVELIN Renal 101)
Pfizer | Phase 3 Completed Jun 26, 2024 | INTERVENTIONAL
| Renal Cell Cancer |
The TransCatheter Valve and Vessels Trial
Medtronic | Phase not applicable Completed Jun 26, 2024 | INTERVENTIONAL
| Aortic Stenosis
CABG
Fractional Flow Reserve
Multi Vessel Coronary Artery Disease
PCI
TAVI |
Safety and Tolerability of the Ophthalmic Gel PRO-165 Versus Artelac® Nightime Gel
Laboratorios Sophia S.A de C.V. | Phase 1 Completed Jun 26, 2024 | INTERVENTIONAL
| Dry Eye Syndromes |
Trial to Evaluate the Safety and Pharmacokinetics of HMPL-689 in Patients With Lymphomas
Hutchmed | Phase 1 Completed Jun 26, 2024 | INTERVENTIONAL
| Lymphoma |
Treatment of GVHD in Hematopoietic Stem Cell Transplant (HSCT) Recipients Using AAT Plus Corticosteroids (CS) Compared With Corticosteroids Alone
CSL Behring | Phase 3 Completed Jun 26, 2024 | INTERVENTIONAL
| Graft Versus Host Disease (GVHD) |
An Open-Label and Long-Term Extension Study to Evaluate the Efficacy and Safety of Ustekinumab in the Treatment of Patients With Ichthyoses
Janssen Scientific Affairs, LLC | Phase 1 Completed Jun 26, 2024 | INTERVENTIONAL
| Ichthyosis |
Asciminib in Monotherapy for Chronic Myeloid Leukemia in Chronic Phase (CML-CP) With and Without T315I Mutation
Novartis Pharmaceuticals | Phase 3 Completed Jun 26, 2024 | INTERVENTIONAL
| Chronic Myelogenous Leukemia - Chronic Phase |
A Study of Abemaciclib (LY2835219) Plus Hormone Therapy in Participants With Early Breast Cancer
Eli Lilly and Company | Phase 3 Completed Jun 26, 2024 | INTERVENTIONAL
| Breast Neoplasms |
Natural History Study of ENPP1 Deficiency and the Early-onset Form of ABCC6 Deficiency
Inozyme Pharma | Phase not applicable Completed Jun 26, 2024 | OBSERVATIONAL
| ATP-Binding Cassette Subfamily C Member 6 Deficiency
Autosomal Recessive Hypophosphatemic Rickets
Ectonucleotide Pyrophosphatase/phosphodiesterase1 Deficiency
Generalized Arterial Calcification of Infancy |
A Study of JNJ-73763989, JNJ-64300535, and Nucleos(t)Ide Analogs in Virologically Suppressed, Hepatitis B e Antigen (HBeAg)- Negative Participants With Chronic Hepatitis B Virus Infection
Janssen Research & Development, LLC | Phase 1 Completed Jun 26, 2024 | INTERVENTIONAL
| Hepatitis B, Chronic |
A Study of JNJ-64281802 for the Prevention of Dengue Infection
Janssen Research & Development, LLC | Phase 2 Completed Jun 26, 2024 | INTERVENTIONAL
| Dengue |
Study of Magrolimab Given Together With FOLFIRI/Bevacizumab (BEV) in Participants With Previously Treated Advanced Inoperable Metastatic Colorectal Cancer (mCRC)
Gilead Sciences | Phase 2 Completed Jun 26, 2024 | INTERVENTIONAL
| Metastatic Colorectal Cancer |
A Human Tick Kill Study to Investigate the Safety, Tolerability, and Whole Blood Concentration of TP-05
Tarsus Pharmaceuticals, Inc. | Phase 2 Completed Jun 26, 2024 | INTERVENTIONAL
| Healthy Volunteer |
A Study of PRT3645 in Participants With Select Advanced or Metastatic Solid Tumors
Prelude Therapeutics | Phase 1 Completed Jun 26, 2024 | INTERVENTIONAL
| Breast Cancer
Endometrial Cancer
Glioblastoma
Head and Neck Squamous Cell Carcinoma
Malignant Mesothelioma
Non-small Cell Lung Cancers
Sarcoma |
Effect of Extruded Cereals Enriched With β-glucan on Postprandial Plasma Glucose and Serum Insulin Responses
Cereal Partners Worldwide | Phase not applicable Completed Jun 26, 2024 | INTERVENTIONAL
| Healthy |
Effect of Extruded Cereals Enriched With β-glucan on Postprandial Plasma Glucose and Serum Insulin Responses
INQUIS Clinical Research | Phase not applicable Completed Jun 26, 2024 | INTERVENTIONAL
| Healthy |
Effect of Extruded Cereals Enriched With β-glucan on Postprandial Plasma Glucose and Serum Insulin Responses
Société des Produits Nestlé (SPN) | Phase not applicable Completed Jun 26, 2024 | INTERVENTIONAL
| Healthy |
Vascular Positioning System G4 Algorithm ECG Data Collection for Model Training Study
Teleflex | Phase not applicable Completed Jun 26, 2024 | OBSERVATIONAL
| Peripheral ICC |
A Study to Learn About a Type of Pneumococcal Vaccine With a New Ingredient (PF-07872411) Intended to Enhance the Effects of the Vaccine Which is Given to Adults
Pfizer | Phase 1 Completed Jun 26, 2024 | INTERVENTIONAL
| Pneumococcal Disease |
Efficacy, Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of ZYIL1 in Patients With Amyotrophic Lateral Sclerosis
Zydus Lifesciences Limited | Phase 2 Completed Jun 26, 2024 | INTERVENTIONAL
| Amyotrophic Lateral Sclerosis |
Fibricheck Detection cApabilities for Atrial Fibrillation
Qompium NV | Phase not applicable Completed Jun 26, 2024 | INTERVENTIONAL
| Atrial Fibrillation |
A Thorough QT Study of ABBV-CLS-7262 in Healthy Subjects
AbbVie | Phase 1 Completed Jun 26, 2024 | INTERVENTIONAL
| Healthy |
A Thorough QT Study of ABBV-CLS-7262 in Healthy Subjects
Calico Life Sciences LLC | Phase 1 Completed Jun 26, 2024 | INTERVENTIONAL
| Healthy |
Prevalence of DAO (diamino Oxidase) Deficiency in Newborns
AB Biotek | Phase not applicable Completed Jun 26, 2024 | No interventions listed | DAO Genetic Variants |
A Study of Doxorubicin Plus Olaratumab (LY3012207) in Participants With Advanced or Metastatic Soft Tissue Sarcoma
Eli Lilly and Company | Phase 3 Completed Jun 27, 2024 | INTERVENTIONAL
| Soft Tissue Sarcoma |
A Study of Anti-PD-L1 Checkpoint Antibody (LY3300054) Alone and in Combination in Participants With Advanced Refractory Solid Tumors
Eli Lilly and Company | Phase 1 Completed Jun 27, 2024 | INTERVENTIONAL
| Breast Cancer (HR+HER2-)
Cutaneous Melanoma
Microsatellite Instability-High (MSI-H) Solid Tumors
Pancreatic Cancer
Solid Tumor |
Assessing the Clinical Benefit of Molecular Profiling in Patients With Solid Tumors
Strata Oncology | Phase not applicable Completed Jun 27, 2024 | No interventions listed | Adult Solid Tumor
Cancer
Lymphoma
Multiple Myeloma |
Study of Ibrutinib Combined With Venetoclax in Subjects With Mantle Cell Lymphoma (MCL)
Janssen Research & Development, LLC | Phase 3 Completed Jun 27, 2024 | INTERVENTIONAL
| Mantle-Cell Lymphoma |
Study of Ibrutinib Combined With Venetoclax in Subjects With Mantle Cell Lymphoma (MCL)
Pharmacyclics LLC. | Phase 3 Completed Jun 27, 2024 | INTERVENTIONAL
| Mantle-Cell Lymphoma |
Sleep To Reduce Incident Depression Effectively
Big Health Inc. | Phase not applicable Completed Jun 27, 2024 | INTERVENTIONAL
| Insomnia, Primary
Major Depressive Disorder
Rumination |
A Study to Test GlaxoSmithKline's (GSK) Herpes Zoster (HZ) Subunit Vaccine's Long-term Immune Response in Previously Vaccinated Kidney Transplant Adults and Then to Test if 2 Additional Doses of the Vaccine Are Safe and Able to Generate an Immune Response
GlaxoSmithKline | Phase 3 Completed Jun 27, 2024 | INTERVENTIONAL
| Herpes Zoster |
A Study of Bevacizumab, Carboplatin, and Paclitaxel or Pemetrexed With or Without Atezolizumab in Chemotherapy-Naïve Patients With Stage IV Non-Squamous Non-Small Cell Lung Cancer (IMpower151)
Hoffmann-La Roche | Phase 3 Completed Jun 27, 2024 | INTERVENTIONAL
| Carcinoma, Non-Small-Cell Lung |
Treatment of Post-Operative Pain Following Orthopedic Surgery With SPRINT® Peripheral Nerve Stimulation (PNS) System
SPR Therapeutics, Inc. | Phase not applicable Completed Jun 27, 2024 | INTERVENTIONAL
| Partial Knee Replacement
Postoperative Pain
Total Knee Arthroplasty
Total Knee Replacement |
Assessment of the Efficacy, Safety, and Tolerability of AVP-786 for the Treatment of Agitation in Patients With Dementia of the Alzheimer's Type
Otsuka Pharmaceutical Development & Commercialization, Inc. | Phase 3 Completed Jun 27, 2024 | INTERVENTIONAL
| Agitation in Patients With Dementia of the Alzheimer's Type |
Multicentric Clinical Study on Isatis TF Trifocal Intraocular Lens
Cutting Edge SAS | Phase not applicable Completed Jun 27, 2024 | INTERVENTIONAL
| Cataract
Lens Opacities |
Multicentric Clinical Study on Isatis TF Trifocal Intraocular Lens
targomedGmbH | Phase not applicable Completed Jun 27, 2024 | INTERVENTIONAL
| Cataract
Lens Opacities |
Safety and Efficacy of NTX-001 Compared to SOC in Acute Single Peripheral Nerve Injuries
Neuraptive Therapeutics Inc. | Phase 2 Completed Jun 27, 2024 | INTERVENTIONAL
| Peripheral Nerve Injuries |
Study Comparing Two Versus Three Active MDMA-assisted Sessions in U.S. Military Veterans With Chronic PTSD
Lykos Therapeutics | Phase 2 Completed Jun 27, 2024 | INTERVENTIONAL
| PTSD |
Clinical Use of EVUSHELD as Pre-exposure Prophylaxis in Real-world Setting in Gulf Cooperation Council Countries
AstraZeneca | Phase not applicable Completed Jun 27, 2024 | No interventions listed | COVID-19 |
A Study to Learn About the Study Medicine (Called Avelumab) in People With Advanced Urothelial Cancer After Chemotherapy
Merck Healthcare KGaA, Darmstadt, Germany, an affiliate of Merck KGaA, Darmstadt, Germany | Phase not applicable Completed Jun 27, 2024 | OBSERVATIONAL
| Urothelial Cancer |
Open-Label Extension Study of ASTORIA
Contera Pharma A/S | Phase 2 Completed Jun 27, 2024 | INTERVENTIONAL
| Dyskinesias |
Active-control Randomized Trial Comparing 4-factor Prothrombin Complex Concentrate With Frozen Plasma in Cardiac Surgery
Octapharma | Phase 3 Completed Jun 27, 2024 | INTERVENTIONAL
| Bleeding Cardiac Surgery Patients |
Clinical Study to Evaluate the Performance of Synthetic Polyurethane Male Condoms
Okamoto Industries, Inc. | Phase not applicable Completed Jun 27, 2024 | INTERVENTIONAL
| Performance of Synthetic Polyurethane Male Condom |
A Study to Investigate the Safety, Tolerability, Immunogenicity, and Pharmacodynamics of VXX-401 Administered IM in Adult Participants
Novotech (Australia) Pty Limited | Active
Phase 1 Past estimated: Jun 27, 2024 | INTERVENTIONAL
| Hypercholesterolemia |
A Study to Investigate the Safety, Tolerability, Immunogenicity, and Pharmacodynamics of VXX-401 Administered IM in Adult Participants
Vaxxinity, Inc. | Active
Phase 1 Past estimated: Jun 27, 2024 | INTERVENTIONAL
| Hypercholesterolemia |
Data Source and Provenance
All data is sourced from the Aggregate Content of ClinicalTrials.gov, put together by the Clinical Trials Transformation Initiative (CTTI). This is a fantastic project and resource that makes the canonical data on ClinicalTrials.gov available for researchers like us. We refresh our clinical trial database with AACT data daily. This data table is a selection of the most recent 500 studies since June 2024.
Page Last Refreshed: Dec 24, 2025